Supervised by: Aparajita Sriram, MSc, MPhil. Aparajita is currently completing an MPhil degree in genomic medicine at the University of Cambridge, and is looking forward to starting a research position at the University of Exeter to look at the genetic basis of rare types of diabetes. She is particularly interested in circadian rhythms and psychiatry.
ABSTRACT
Colorectal cancer (CRC) persists as an alarming and formidable global health burden, ranking as the third most common type of cancer worldwide and taking 0.9 million lives in 2020. Colorectal cancer typically arises from the uncontrolled growth of abnormal cells in the lining of the colon or rectum, often starting as benign polyps that can eventually become cancerous if left untreated, growing at a relatively slow rate. Several individual risk factors contribute to the development of CRC, including age (most cases occur after age 50), family history of CRC or certain genetic syndromes, personal history of inflammatory bowel disease (such as Crohn’s disease or ulcerative colitis), and other environmental risk factors. Due to the disease’s slow growth, early detection through effective screening methods is crucial for reducing CRC-related fatalities. There are currently various options for CRC screening including faecal occult blood tests, sigmoidoscopies, colonoscopies, DNA stool tests and genetic biomarkers. Each of these tests have their own set of advantages and disadvantages, with the faecal immunochemical test (FIT) emerging as the most preeminent approach in CRC screening worldwide. However, CRC screening also has some disadvantages. Invasive procedures like colonoscopy can be uncomfortable, carry a small risk of complications, and false positives can lead to unnecessary anxiety and additional tests. Risk stratified screening could enable patients to receive individualised care corresponding to their level of risk, receive an early diagnosis that enhances health outcomes, and choose further procedures more wisely by avoiding unnecessary operations, thus improving the benefit-to-harm ratio. This literature review examines the benefits and perils of various CRC screening strategies, as well as risk stratification in CRC screening.
INTRODUCTION
Colorectal cancer (CRC), a significant global health burden, originates in the colon or rectum due to intricate genetic mutations and environmental influences. This malignancy presents with a range of clinical manifestations, including alterations in bowel habits, rectal bleeding, abdominal pain, and unintentional weight loss, making early detection pivotal.
The prevalence of CRC is significantly high in developed countries such as the United States, Canada, Australia, and many European nations when compared to developing or underdeveloped countries. People over the age of 50 at average risk of CRC are often recommended for regular screening by governments and healthcare organisations. In the United States there is a 4.4% chance of a man’s lifetime prevalence of CRC and a 4.1% chance of a woman’s lifetime prevalence of CRC. (Rawla, 2019)
The number of colorectal cancer cases in the United States has been decreasing since the mid-1980s. During the 2000s, incidence rates dropped 3% to 4% each year. This was due to increased screening in adults aged 50 and older. From 2011 to 2019, incidence rates continued to decrease by 1% annually. (“Colorectal cancer”, 2022)
Risk factors associated with colorectal cancer
Colorectal cancer is associated with nonmodifiable risk factors, including age and hereditary factors, as well as modifiable factors related to the environment and lifestyle.
Age
The chance of developing cancer increases after age 50, and nine out of ten people who develop colorectal cancer are over 50 years old. (Lewandowska, 2022) However, colorectal cancer appears to be increasing among young people. In fact, in the US, colorectal cancer is now one of the ten most commonly diagnosed cancers among persons 20 to 49 years (de Jong, 2005).
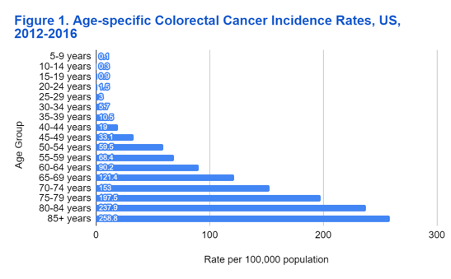
Figure 1: Age Specific CRC Incidence Rates, US 2012-16. (NAACCR, 2019)
Personal History of Adenomatous Polyps and Inflammatory Bowel Disease
Neoplastic polyps of the colorectum are precursor lesions of colorectal cancer. An adenoma is a benign (noncancerous) tumour. A person with a history of adenomas has a greater risk of developing cancer than persons with no previous history of adenomas. ( Janout, 2001) First-degree relatives of patients with newly diagnosed adenomas are at increased risk for colorectal cancer. (Johnson, 2007)
Inflammatory bowel disease (IBD) is a term used to describe two diseases, Ulcerative colitis and Crohn disease. These conditions increase an individual’s risk of developing colorectal cancer. (Davies, 2005) The risk of CRC in patients with inflammatory bowel disease is 4 to 20-fold. (Lynch, 2008)
Inherited Genetic Risk
5 to 10% of colorectal cancers are caused by inherited genetic conditions. The most common inherited diseases include Familial Adenomatous Polyposis (FAP) and Hereditary Nonpolyposis Colorectal Cancer (HNPCC), also called Lynch syndrome. Lynch syndrome is characterized by germline inactivation of one allele of genes involved in the mismatch repair system, namely MLH1, MSH2, MSH6, PMS2, or EPCAM. (Haggar, 2009)
The lifetime risk of colorectal cancer in people with recognized HNPCC-related mutations is as high as 70 to 80%. FAP, on the other hand, is caused by mutations in the tumour suppressor gene APC. Unlike individuals with HNPCC, who develop only a few adenomas, people with FAP develop hundreds, usually at a young age, and one or more of these adenomas typically undergoes a malignant transformation as early as age 20(McMichael, 1980). By age 40, almost all people with this disorder will have developed cancer if the colon is not removed. Approximately 75 to 80% of individuals with APC-associated polyposis conditions have an affected parent. Prenatal testing and preimplantation genetic diagnosis are possible if a mutation is identified in an affected family member (‘WCRF’, 2007).
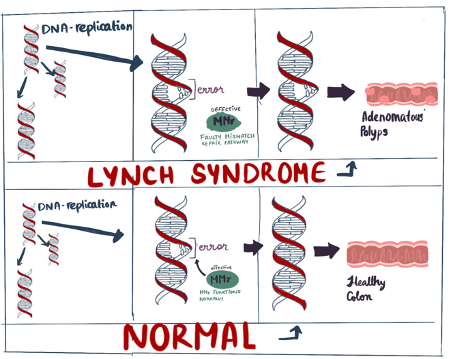
Figure 2: Simplified Lynch Syndrome Tumorigenesis pathway. (Ibrahim, 2023)
Environmental Risk Factors
Environmental factors are defined broadly to include a range of often ill-defined cultural, social, and lifestyle factors. Colorectal cancer is widely considered to be highly dependent on environmental factors. Strong scientific evidence shows that obese people have a greater risk of developing colorectal cancer and that risk increases with increasing BMI. (de Jong, 2005)
Diets high in animal fat are a major risk factor for colorectal cancer. Smoking and alcohol consumption are also risk factors for CRC. Evidence shows that 12% of colorectal cancer deaths are attributed to smoking. (Zisman, 2006) The carcinogens found in tobacco increase cancer growth in the colon and rectum. Alcohol consumption is a factor in the onset of colorectal cancer at a younger age as well as a disproportionate increase of tumours in the distal colon. Interestingly, some evidence of environmental risk arises from studies of migrants and their offspring. Among migrants from low risk to high-risk countries, incidence rates of colorectal cancer tend to increase towards those typical of the population of the host country. (‘World Cancer Research Fund and American Institute for Cancer Research’, 2007).
Screening Tests for colorectal cancer
When an individual does not exhibit any symptoms of a condition, a screening test is performed to look for probable health issues or diseases. Considering colon cancer is the third most common cancer globally, early diagnosis and treatment, through screening, can increase survival by 5 and 10 years, and by more than 50% in younger patients. Hence there is a need for good screening that allows early detection in high-risk groups. (‘Colorectal Cancer – Screening’, 2022)
There are different types of screening tests for CRC, like- faecal occult blood test (FOBT) and faecal immunochemical test (FIT)- which are used to detect blood in the faeces, double contrast barium enema (DCBE)- which involves consumption of enema containing barium that brightens colon during x-ray scan, sigmoidoscopy- which checks for polyps/ abnormalities in the rectum and lower colon only, and stool DNA test- which involves analysing changes of DNA in an individual’s stool sample. Each of these tests have their own advantages, disadvantages, and a frequency at which they should be conducted. (‘Colorectal Cancer – Screening’, 2022)
Diagnostic tests for colorectal cancer
Individuals who have a ‘positive screening test’ or those who have symptoms strongly suggestive of colorectal cancer are urged to conduct diagnostic tests. The types of diagnostic tests available are- colonoscopy (colonoscopist checks the entire rectum and colon for cancer), biopsies and pathological examination, molecular tests (to identify unique genetic features of the tumour- such as, defects in mismatch repair genes), blood tests, as well as imaging techniques such as CT scan, MRI, Ultrasound, X-ray and PET- CT scan to detect the size, structure and localization of the tumour. (‘Colorectal Cancer – Diagnosis’, 2022)
Treatment of colorectal cancer
If the diagnosis is CRC, different treatment options can be considered, including- surgery (laparoscopic surgery, colostomy, cryoablation), radiation therapy (external- beam, stereotactic, intraoperative, brachytherapy etc.) and medication (chemotherapy, targeted therapy, immunotherapy). (‘Colorectal Cancer – Types of Treatment’, 2023)
REVIEW OF LITERATURE
Colorectal Cancer Screening Strategies
Faecal occult blood test (FOBT)
The Faecal Occult Blood Test (FOBT) is a screening method for detecting hidden blood in stool, which may indicate colorectal cancer. It involves collecting a small stool sample at home or a medical facility and sending it to a lab for analysis. (“Faecal Occult Blood Tests”, 2021)The advantage is that it’s a simple screening method that can help detect early signs of colorectal cancer, potentially leading to timely interventions and improved outcomes. However the disadvantage is that FOBT has a higher false-positive rate compared to other screening tests, leading to unnecessary anxiety and nervousness (Hewitson, 2007).
Faecal Immunochemical Test (FIT)
FIT (Faecal Immunochemical Test) is a simple, non-invasive way to detect potential colorectal issues by performing a simple, non-invasive procedure. Patients can perform the test at home without the need for any special preparation or dietary restrictions. The procedure involves collecting a small stool sample and placing it on a card or into a container provided by the healthcare provider. The sample is then sent to a laboratory for analysis, where it is checked for the presence of blood, which could be an early sign of colorectal cancer or other gastrointestinal conditions. If the test comes back positive for blood, it may indicate the need for further evaluation, such as a colonoscopy, to confirm or rule out any serious concerns. FIT allows patients to take an initial step towards screening without immediately subjecting them to intense or invasive testing, making it a more accessible and patient-friendly option for early detection. Regular screenings can significantly improve the chances of detecting and treating colorectal issues in their early stages, leading to better outcomes and reduced mortality rates (Song, 2016).
Colonoscopy
Colonoscopy is a medical procedure used to examine the inner lining of the large intestine (colon) and rectum. It is primarily employed to detect and prevent colorectal cancer and diagnose various gastrointestinal conditions. During the procedure, a flexible, thin tube with a camera at its end, called a colonoscopy, is inserted through the rectum into the colon. This allows the doctor to visually inspect the colon for abnormalities like polyps or inflammation. (Stauffer, 2023) One pro is Colonoscopy is a highly effective and minimally invasive method for detecting and removing precancerous growths, potentially preventing the development of colorectal cancer. However the procedure requires thorough bowel preparation, which can be uncomfortable and time-consuming for some patients, and it is associated with risks such as perforation of the colon, bleeding, severe pain, reactions to sedatives, and even death (which is very rare). (Keum, 2021)
Flexible sigmoidoscopy
Flexible sigmoidoscopy is a minimally invasive medical technique used to look for anomalies in the rectum and lower section of the colon. A flexible tube (sigmoidoscope) containing a light and camera is introduced into the anus and progressed through the colon during the operation. Real-time photos from the camera can be used to find polyps, inflammation, and other problems (‘Flexible Sigmoidoscopy’, 2016).
Stool DNA test
Stool DNA test, also known as a colorectal cancer screening test, is a non-invasive method to detect abnormal DNA changes in the stool that may indicate the presence of colorectal cancer or precancerous growths. During the procedure, a small sample of stool is collected at home and sent to a lab for analysis. One advantage of the test is its convenience, as it eliminates the need for uncomfortable procedures like colonoscopies. However, a drawback is its potential for false positives and false negatives, leading to unnecessary anxiety or missed detections. Regular screenings and consultation with healthcare professionals are essential for accurate results and proper follow-up (Abbaszadegan, 2007). Stool DNA analysis is used to detect genetic abnormalities associated with colorectal cancer and other gastrointestinal conditions. False positives can occur due to non-cancerous sources of DNA in the stool, while false negatives can result from low levels of abnormal DNA or sampling errors.
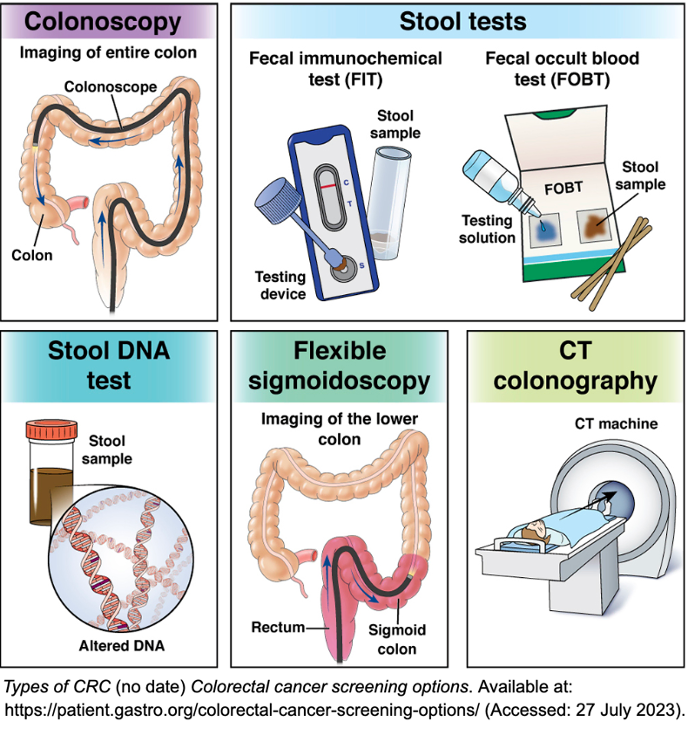
Figure 3: Colorectal cancer screening options. (Available at https://patient.gastro.org/colorectal-cancer-screening-options/)
Genetic biomarkers
Genetic biomarkers for colorectal cancer (CRC) are specific DNA alterations used to aid in diagnosis and prognosis. The procedure involves analysing the patient’s genetic material, usually through a blood or tissue sample, to detect mutations associated with CRC (Tanaka, 2019). One advantage of using genetic biomarkers is their high sensitivity and specificity, enabling early detection and personalised treatment plans. However, a disadvantage is that not all biomarkers may be well understood, leading to potential misinterpretations and limited treatment options. Additionally, the cost and accessibility of genetic testing could pose challenges for widespread implementation, hindering equitable healthcare access (Coppedè, 2014). The table below outlines the key gene variants associated with hereditary syndromes and their roles in colorectal cancer.
| Gene | Hereditary Syndrome | Population Frequency | Role in CRC |
| APC | Familial Adenomatous Polyposis (FAP) | 0.01-0.02% | Tumour suppressor gene; frequently mutated in familial and sporadic CRC |
| MSH2/ MSH6/ MLH1/ PMS2 | Lynch Syndrome (Hereditary Nonpolyposis Colorectal Cancer, HNPCC) | <0.10% | DNA mismatch repair genes; mutations lead to microsatellite instability and increased CRC risk |
| BMPR1A | Juvenile Polyposis Syndrome (JPS) | <0.1% | Tumour suppressor gene; involved in TGF-β signalling pathway; mutations associated with juvenile polyposis and CRC |
| SMAD4 | Juvenile Polyposis Syndrome (JPS) | <0.5% | Tumour suppressor gene; involved in TGF-β signalling pathway; mutations in advanced CRC |
Table 1: Gene Variants and Their Roles in Colorectal Cancer (CRC) and Hereditary Syndromes (Bray, 2007), (Gut, 2013)
However, screening tests can do more harm than good when administered to individuals who are not at a high risk for disease or when testing for extremely rare disorders. A few of the disadvantages of screening include:
- Incorrect results: A screening test could give rise to false negative and false positive results. A false negative result is when someone with a negative screening result has the target condition, while a false positive result is when someone with a positive screening result does not have the target condition.
- Risks associated with the procedure: Few screening tests, like colonoscopy may have side effects, such as perforation of the colon, bleeding, severe pain, reactions to sedatives, and even death (which is very rare).
- Anxiety: This may be caused while conducting invasive screening procedures False positive results of screening tests can also lead to a lot of unnecessary anxiety and psychological harm.
- Over-screening: Some people may misinterpret screening as prevention of disease, resulting in frequently conducting the test for slight abnormal changes which may not be detrimental.
- Discomfort: For example, extensive preparation of preparing the colon and dieting before the screening test, or the discomfort, be it physical or psychological, caused by the screening procedure itself. (‘Colorectal Cancer – Screening’, 2022)
Comparison between various colorectal cancer screening tests
I. Cost-effectiveness and Affordability
In healthcare, cost-effectiveness best refers to the assessment of the value gained from medical interventions in relation to their associated costs. This involves analysing the effectiveness of certain healthcare strategies, treatments, or preventive measures to achieve a desired health outcome while considering the financial resources expended. The importance of cost-effectiveness in promoting the widespread adoption of CRC screening lies in its ability to demonstrate the efficiency and impact of screening programs (Thomas and Chalkidou, 2016). By evaluating the cost-effectiveness of screening tests, healthcare providers can make informed decisions, ensuring that resources are directed towards interventions that offer the greatest health benefits for the most reasonable cost which directly makes CRC screening more accessible and financially feasible for both healthcare systems and individuals.
Comparing the costs and cost-effectiveness of different CRC screening tests requires various factors, such as their effectiveness in detecting colorectal cancer, the requirement of follow-up procedures, and the overall impact on healthcare resources (Lansdorp-Vogelaar, 2010).
Colonoscopy
Colonoscopies are generally more expensive than other screening tests due to the test’s invasiveness and the need for specialised equipment and trained personnel. Despite this, colonoscopies are generally highly effective in detecting colorectal cancer and precancerous lesions, making it a preferred choice for comprehensive screening. If polyps or suspicious lesions are found during the procedure, follow-up procedures like a biopsy or polypectomy may be necessary.
FIT/FOBT
FOBT and FIT on the other hand are more cost-effective compared to colonoscopy because they are non-invasive and do not require specialised equipment. While FOBT and FIT are effective in detecting blood in stool, they may have lower sensitivity for early-stage colorectal cancer and smaller polyps compared to methods such as colonoscopy (Knudsen, 2010). Positive FOBT/FIT results typically necessitate additional diagnostic evaluations, such as a colonoscopy, to confirm the presence of colorectal cancer or precancerous lesions. FOBT and FIT are relatively simple and less resource-intensive, making them more accessible and efficient in utilising healthcare resources.
Stool DNA tests
Stool DNA tests are typically more expensive than FOBT and FIT but less expensive than a colonoscopy due to the use of advanced molecular technologies for detecting genetic changes associated with colorectal cancer. Stool DNA tests have shown promise in detecting both colorectal cancer and advanced adenomas, offering higher sensitivity than traditional FOBT/FIT (Brenner, 2010). Positive stool DNA test results typically require follow-up with colonoscopy for further evaluation and confirmation. These tests are also more resource-intensive compared to FOBT and FIT, but their higher sensitivity can lead to earlier detection, potentially reducing the need for subsequent screening tests.
Healthcare systems must carefully consider these factors when designing screening programs to ensure the maximisation of early detection and prevention of colorectal cancer.
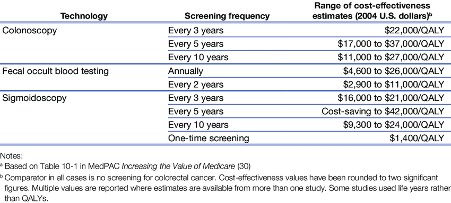
Table 2: Cost-effectiveness of alternative colorectal cancer screening interventions (Naber, 2019)
II. Accessibility and Feasibility
Accessibility in this context pertains to the availability of medical screening procedures to all population groups, e.g., people of differing socio-economic backgrounds. Feasibility refers to how simply a procedure can be operated and is determined by the amount of responsibility of an individual during screening. A good understanding of both accessibility and feasibility are vital in making informed differentiations between screening procedures that are important to consider during a patient’s decision-making process. This also allows for healthcare systems to examine screening procedures’ strengths as well as inspect limitations – allowing room for improvement and encouraging a patient’s autonomy in choosing the most appropriate option suited to their personal needs. Also, success of screening programs is heavily dependent on public perception and social awareness regarding healthcare. Hence, an analysis of the sub-topic above can play a crucial role in healthcare-related policy-making processes carried out by governments worldwide.
Comparing the accessibility and feasibility of different CRC screening tests requires discussion concerning screening promotion plans initiated by the government; the simplicity of the procedure; the capacity to which people of all age-groups as well as people with any disabilities can participate in CRC screening tests.
Colonoscopy
Colonoscopies are more expensive compared to other CRC screening methods, making it less accessible to people from a low-income background. Although people with health insurance are relieved significantly from costs, accessibility is greatly reduced as only people with finances to spare for screening will participate. Moreover, colonoscopies involve insertion of a colonoscope into one’s rectum for examination. One’s concern regarding privacy or feelings of embarrassment is one of the leading reasons for avoidance of colonoscopy.(Pluskota, 2023)
Colonoscopies are also usually resource-intensive and are one of the most sensitive, thorough CRC screening methods. This makes it less feasible, and a more challenging type of screening for healthcare systems to implement. Hence it is also not an easy task to encourage people to participate in colonoscopies or offer an incentive for them to do so.
However, the concept of ‘Open Access Colonoscopies’ that has arisen in recent years does not require the patient to have an office visit gastroenterologist prior to their procedure. The exchange of all needed information is done over the phone, saving the patient both time and the expense of an office visit. This means that colonoscopies could be much more accessible and feasible on a large scale to qualifying patients. (Havranek, 2020)
FIT/FOBT
FIT is one of the most popular screening methods due its non-invasive nature – meaning that there is no introduction of medical instruments into the human body for usage. This ensures maximum participation of individuals in colorectal cancer screening as it does not breach privacy and involves minimal discomfort.
Most FITs only require 1 or 2 stool samples, and none require dietary, lifestyle, or medication restriction, increasing ease of use. Because of this, countries in Europe and Asia have adopted widespread CRC screening programs using FITs. (‘Ann Intern Med’, 2014)
Moreover, FITs are inexpensive, and can be carried out at the privacy of one’s home, making it often more preferable than a colonoscopy or other doctor-operated procedures. However, diagnostic characteristics of FIT are difficult to estimate, with reported sensitivity ranging from 25%-100%. This lack of precise estimate of sensitivity has resulted in confusion in the healthcare community regarding the causes of this, and ultimately the reliability of FIT. (‘J Gastrointest Oncol’, 2019) Additionally, FIT tests are likely to perform poorly without refrigeration in warm climates and their quality can be affected by postal delays – meaning there are numerous instances where the sensitivity and specificity can be negatively impacted by degradation of an FIT. (‘Sci Rep’, 2019)
Furthermore, the FIT is most effective when repeated each year (‘CMAJ’, 2020), but this repetition often appears to be unnecessary in the eyes of the public, reducing overall efficacy of the FIT in reducing CRC fatalities. It also means that there is a higher probability of a false positives/negatives in the span of years the FIT is taken.
Sigmoidoscopy
Compared to a colonoscopy, a sigmoidoscopy is generally deemed as safer. It is also less complex, and much cheaper (less resource-intensive). As a sigmoidoscopy only looks at the lower portion of one’s colon, there is less of a chance for bleeding or perforation due to any abrasion (Mayo Clinic, 2022). Hence, it is an accessible as well as feasible option for those who need more thorough screening than an FIT, but a less expensive and invasive screening than a colonoscopy.
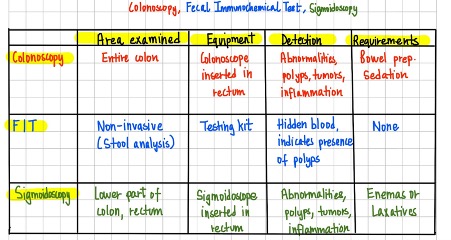
Table 3: Differences between Colonoscopy, FIT, and Sigmoidoscopy
The table above compares different types of CRC screening in terms of area examined (invasiveness), equipment used (resource intensity), means of detection, and requirements prior screening.
III. Accuracy and Sensitivity
Accuracy can best be defined as the ability of a screening test to produce results that are close to the true condition the test aims to detect. When applied to CRC screening, accuracy can determine how well a certain type of test correctly identifies individuals who either have or do not have colorectal cancer. An accurate screening test is mainly effective at minimising both false positives and false negatives (Lee, 2014). Sensitivity, also known as the true positive rate or recall, measures the ability of a screening test to correctly identify individuals who have colorectal cancer among those who do have the disease. It is an essential indicator of the test’s ability to detect existing cases of cancer accurately. A high sensitivity value indicates that the test is effective at recognizing most individuals with CRC (Lin, 2016). High accuracy and sensitivity are crucial factors in ensuring reliable screening results in the context of CRC screening as they are both directly linked to early detection, improved patient outcomes, and efficient resource allocation. Early detection allows these screening tests to identify colorectal cancer in its initial stages, often before symptoms are seen. This early identification enables prompt medical intervention when treatment options are most effective, leading to improved patient outcomes and increased chances of successful treatment (Rutter, 2018). Additionally, reliable screening results aid in efficient resource allocation by directing healthcare resources and interventions to those who genuinely need them, optimising the use of medical facilities, personnel, and financial resources for maximum benefit.
Implementation of national CRC screening programs
The United States
In the United States of America, there are various attitudes towards CRC screening amongst the population including the public, government, and healthcare professionals. While many individuals understand the importance of screening for early cancer detection, there is still a considerable portion of the population that remains hesitant or unaware of the benefits of CRC screening. Screening may be avoided due to fear, misconceptions about the procedure, or a lack of awareness about the risk for colorectal cancer (Bynum, 2012).There are a series of barriers and challenges that can be seen as present within the U.S. including lack of health insurance coverage or high out-of-pocket costs and limited access to healthcare or awareness.
Despite a series of challenges, The U.S. government has implemented several initiatives to promote CRC screening and address these barriers. For example, the Centers for Disease Control and Prevention (CDC) has launched programs such as the Colorectal Cancer Control Program (CRCCP), which provides funding to states and tribal organisations to increase CRC screening rates among low-income, uninsured, and underinsured populations (Hannon, 2013). The program states that its purpose is to “increase colorectal cancer screening rates among people between 45 and 75 years of age.”Through this and other programs, the United States continues to work towards improving accessibility and equitable access to CRC screening programs. These efforts are essential in reducing disparities in colorectal cancer outcomes and increasing the early detection of the disease, ultimately leading to improved patient outcomes and better
The United Kingdom
Key cancer screening programs are provided free of charge in the UK. However individuals must meet specific eligibility criteria, such as age ranges, to be invited to participate in respective screening programs. The NHS Bowel Cancer Screening Program was rolled out in 2006 to increase early detection and improve outcomes for bowel cancer patients in the United Kingdom. It is available to everyone aged 60-74 years. Everyone in this age group who is registered with a General Practitioner is automatically sent an FIT kit every 2 years. (‘Cancer Research UK’, 2021)
The FIT kit was opted for by the government because of its convenience – it can be performed at home by simply collecting a small stool sample and sending it to a screening centre for analysis. The process of screening as well as instructions for usage are explained in a letter from the NHS, and are also readily available online. The NHS and related health authorities run public awareness campaigns (such as the Help Us Help You campaign launched in October 2018) to inform the target population about the importance of bowel cancer screening. These campaigns aim to increase participation rates and ensure people understand the benefits of early detection (‘NHS’, 2020).
Risk Stratification
Risk stratification is the process of systematically categorising a group of patients based on a combination of objective and subjective data, coming up with an assigned risk status to each patient. (Dera, 2019) Healthcare organisations may take into account a variety of factors when determining individual risk levels including advanced age, physical limitations, genetics, or a recent major surgery (Vogelaar, 2022) This data is then used by practices to make care management decisions, typically providing greater access to resources to patients assigned with higher risk categories. A more thorough and intense testing path is usually undertaken by individuals at high risk, while those in the lower-risk group undergo a more lightweight testing process.
Risk stratification aims to assist patients in achieving the optimal health and quality of life by preventing chronic illnesses, progression into higher-risk categories, and further costs. Specifically, CRC risk stratified screening may be able to inform patients about their personal colorectal cancer risk level, which will contribute to the enhancement in patient compliance and attendance for further procedures. It was found in a systematic review published in 2015 that the implementation of risk assessment tools to improve patient knowledge, understanding of the value of screening, and the awareness of risk levels. Findings from 11 randomised trials of 7 cancer risk tools, including 4 trials of CRC risk assessment at a clinical level on 7677 patients were incorporated in the review (Walker, 2015). These findings are also consistently demonstrated in another study: patients in the decision-aid group were more likely to complete a screening test than control patients (43.1% vs 34.8%, p=0,046) within 12 months after the study visit. (Schroy, 2012)
However, opinions on the implementation of risk-stratified screening vary among people and healthcare workers. Supporters argue that risk stratification offers a targeted and cost-effective approach that benefits all patients, clinicians, and service providers. Patients are able to receive individualised care corresponding to their level of risk, receive an early diagnosis that enhances health outcomes, and choose further procedures more wisely by avoiding unnecessary operations, thus improving the benefit-to-harm ratio. Clinicians are able to provide patients with an improved diagnosis and advanced advice on their requests for further screening procedures, while service providers are able to use resources and deliver services more effectively, leading to improved patient compliance. (Hull, 2020)
Critics, on the other hand, may express concerns about potential disparities in access to screening and resources, fearing that individuals in certain socio-economic or geographic groups might be disproportionately affected. Striking a balance between efficient healthcare allocation and equitable access to screening services is crucial, and careful implementation and continuous evaluation of the system’s impact are vital to ensure that risk stratification benefits all individuals regardless of their risk status. Ultimately, a well-designed and inclusive risk stratification system could have the potential to revolutionise CRC screening and potentially serve as a model for other countries seeking to optimise their healthcare strategies.(Hull, 2020) Overall there will always be two sides to consider when a new innovation or idea has come to light, and it is important to understand and analyse both sides to come to an equal yet beneficial conclusion.
The idea of implementing risk stratification into screening programs also raises multiple ethical issues that must be considered in order to ensure the patients’ well-being and human rights. The Emergency Department Technician Handbook introduces the four main principles of medical ethics: beneficence, nonmaleficence, justice, and autonomy, which can be associated with CRC risk stratified screening.
| Medical Ethics | Definition |
| Beneficence | to do good and benefit the patients and society as a whole |
| Nonmaleficence | to do no harm or prevent harm from happening |
| Justice | to treat all patients fairly and equally |
| Autonomy | to allow patients make their own decisions freely |
Table 4: Principles in medical ethics (Shesser, 2023)
Beneficence and nonmaleficence
The ethical concerns with the principles of beneficence and nonmaleficence typically centre on ensuring that the risk assessment procedure promotes well-being and prevents harm. As risk stratified screening for colorectal cancer usually employs population-level risk assessments, it could easily neglect the particular medical condition of each individual patient, meaning that the healthcare decisions that are made might not be able to take specific needs into account. The accuracy and reliability of risk assessment models could also compromise the principle of nonmaleficence. There is always a possibility of individuals receiving inaccurate results, such as false-negatives or false-positives. This could lead to unnecessary and excessive interventions or insufficient preventive procedures that might end up causing more harm than good. Lastly, the concept of providing wider access to resources to higher-risk patients could cause unfair distribution of resources. This could prevent underprivileged people from accessing essential resources, which clearly goes against the principle of beneficence.
Justice
The concept of risk stratified screening underlines the idea of providing greater access to resources to higher-risk patients, as they are considered as the ones who are most in need. This concept however, could easily lead to an unfair distribution of resources. As the majority of insufficient resources are provided to those who are at higher risk, it could prevent underprivileged people from accessing essential resources, which clearly goes against the principle of justice.
Autonomy
The process of risk stratification could directly contradict the principle of autonomy. Once individuals provide their data for the procedure, they may only have limited control over how it is being used. If this lack of control is used beyond the individual’s consent, autonomy may be compromised. The lack of transparency of risk stratification algorithms could also lead to the inability to exercise autonomy over one’s data. The concept of individual freedom may be in conflict if people are unable to fully or even partially understand how their data is being used during the processes.(Dennison 2023) Additionally, it may restrict patients’ choice to select additional desired procedures if medical decisions are made without taking each individual’s particular preferences into account.
CONCLUSION
Due to its high incidence, prevalence, and mortality rate worldwide, colorectal cancer (CRC) has become a global public health problem. Here, we have reviewed the latest discoveries in the study of CRC research and screening and also discussed the merits and demerits of implementing risk stratification into the current screening programs for CRC, with a strong focus on medical ethics. As reviewed above, the onset and development of CRC is induced by a combination of genetic and environmental factors, whose study is essential for the establishment of new prevention strategies. Over the last decade, the major CRC genes have been identified and their pathogenic variants have been associated with a high susceptibility to CRC. Similarly, the development and implementation of new specific and sensitive biomarkers in the foreseeable future will improve diagnostic strategies, thus allowing clinicians to detect CRC cases in the earliest stages of the disease and hence improve the prognosis of thousands of patients. In the context of screening or treatment, personalised medicine is fast becoming an indispensable tool. Risk stratification in colorectal cancer (CRC) testing is a crucial concept that aims to tailor screening approaches based on individual risk levels. Finally, an extensive part of current research into CRC is focused on the development of new therapies and screening strategies that are less aggressive and more effective than conventional ones. Discoveries in this area and their clinical implementation will improve the overall survival and quality of life of CRC patients in the future.
BIBLIOGRAPHY
Abbaszadegan, M.R., Tavasoli, A., Velayati, A., Sima, H.R., Vosooghinia, H., Farzadnia, M., Asadzedeh, H., Gholamin, M., Dadkhah, E. & Aarabi, A. (2007) Stool-based DNA testing, a new noninvasive method for colorectal cancer screening, the first report from Iran. World Journal of Gastroenterology : WJG. 13 (10), 1528–1533. doi:10.3748/wjg.v13.i10.1528.
Ahsan, H., Neugut, A. I., Garbowski, G. C., Jacobson, J. S., Forde, K. A., Treat, M. R., Waye, J. D. (1998). Family history of colorectal adenomatous polyps and increased risk for colorectal cancer. Annals of internal medicine, https://doi.org/10.7326/0003-4819-128-11-199806010-00006
Anon (n.d.) A Systematic Rapid Evidence Assessment of Late Diagnosis. https://eppi.ioe.ac.uk/cms/Default.aspx?tabid=3367#:~:text=Delayed%20diagnosis%20results%20in%20serious,other%20conditions%20is%20less%20clear. [Accessed: 29 July 2023a].
Anon (2012a) Colorectal Cancer – Diagnosis. 25 June 2012. Cancer.Net. https://www.cancer.net/cancer-types/colorectal-cancer/diagnosis [Accessed: 29 July 2023].
Anon (2015) Colorectal Cancer – Screening. 5 October 2015. Cancer.Net. https://www.cancer.net/cancer-types/colorectal-cancer/screening [Accessed: 29 July 2023].
Anon (2012b) Colorectal Cancer – Statistics. 25 June 2012. Cancer.Net. https://www.cancer.net/cancer-types/colorectal-cancer/statistics [Accessed: 29 July 2023].
Anon (2012c) Colorectal Cancer – Types of Treatment. 25 June 2012. Cancer.Net. https://www.cancer.net/cancer-types/colorectal-cancer/types-treatment [Accessed: 29 July 2023].
Anon (2023a) Colorectal Cancer Control Program (CRCCP) | CDC. 13 March 2023. https://www.cdc.gov/cancer/crccp/index.htm [Accessed: 29 July 2023].
Anon (2023b) Colorectal Cancer Screening Tests | CDC. 23 February 2023. https://www.cdc.gov/cancer/colorectal/basic_info/screening/tests.htm [Accessed: 29 July 2023].
Anon (n.d.) Fecal Immunochemical Test. Colorectal Cancer Alliance. https://www.ccalliance.org/screening-prevention/screening-methods/fecal-immunochemical-test [Accessed: 29 July 2023b].
Anon (n.d.) Fecal Occult Blood Test (FOBT): MedlinePlus Medical Test. https://medlineplus.gov/lab-tests/fecal-occult-blood-test-fobt/ [Accessed: 29 July 2023c].
Anon (2019) Screening Tests for Common Diseases. 14 August 2019. https://www.hopkinsmedicine.org/health/treatment-tests-and-therapies/screening-tests-for-common-diseases [Accessed: 29 July 2023].
Anon (2021) Stool Tests for Colorectal Cancer: Types, Pros and Cons. 12 March 2021. City of Hope. https://www.cancercenter.com/cancer-types/colorectal-cancer/diagnosis-and-detection/lab-tests/stool-tests [Accessed: 29 July 2023].
Anon (n.d.) The pros and cons of screening. GOV.UK. https://www.gov.uk/guidance/the-pros-and-cons-of-screening [Accessed: 29 July 2023d].
Anon (n.d.) What Is An REA? – Center for Evidence Based Management. https://cebma.org/faq/what-is-an-rea/.
Anon (n.d.) What Is Colorectal Cancer? | How Does Colorectal Cancer Start?https://www.cancer.org/cancer/types/colon-rectal-cancer/about/what-is-colorectal-cancer.html [Accessed: 29 July 2023f].
Anon (n.d.) Why Do People Avoid Colonoscopies? https://www.uhhospitals.org/blog/articles/2023/05/why-do-people-avoid-colonoscopy [Accessed: 29 July 2023g].
Bynum, S.A., Davis, J.L., Green, B.L. & Katz, R.V. (2012) Unwillingness to participate in colorectal cancer screening: Examining fears, attitudes, and medical mistrust in an ethnically diverse sample of adults 50 years and older. American journal of health promotion : AJHP. 26 (5), 10.4278/ajhp.110113-QUAN-20. doi:10.4278/ajhp.110113-QUAN-20.
Cancer, | (2013) Colonoscopy vs Sigmoidoscopy. 28 March 2013. Scripps Health. https://www.scripps.org/news_items/4457-what-is-the-difference-between-a-colonoscopy-and-a-sigmoidoscopy [Accessed: 29 July 2023].
Coppedè, F., Lopomo, A., Spisni, R. & Migliore, L. (2014) Genetic and epigenetic biomarkers for diagnosis, prognosis and treatment of colorectal cancer. World Journal of Gastroenterology : WJG. 20 (4), 943–956. doi:10.3748/wjg.v20.i4.943.
Davies, R. J., Miller, R., & Coleman, N. (2005). Colorectal cancer screening: prospects for molecular stool analysis. Nature reviews. Cancer https://doi.org/10.1038/nrc1569
de Jong, A. E., Morreau, H., Nagengast, M. H., Mathus-Vliegen, H. M. E., Kleibeuker, H. J., Griffioen, G., Cats, A., Vasen, A. F. H.(2005). Prevalence of adenomas among young individuals at average risk for colorectal cancer. The American Journal of Gastroenterology https://doi.org/10.1111/j.1572-0241.2005.41000.x
Deo, S.V.S., Kumar, S., Bhoriwal, S., Shukla, N.K., Sharma, A., Thulkar, S., Das, P., Bhagat, P., Dhall, K., Pathy, S. & Mohanti, B.K. (2021) Colorectal Cancers in Low- and Middle-Income Countries—Demographic Pattern and Clinical Profile of 970 Patients Treated at a Tertiary Care Cancer Center in India. JCO Global Oncology. (7), 1110–1115. doi:10.1200/GO.21.00111.
Goede, S.L., Rabeneck, L., van Ballegooijen, M., Zauber, A.G., Paszat, L.F., Hoch, J.S., Yong, J.H.E., Kroep, S., Tinmouth, J. & Lansdorp-Vogelaar, I. (2017) Harms, benefits and costs of fecal immunochemical testing versus guaiac fecal occult blood testing for colorectal cancer screening. PLoS ONE. 12 (3), e0172864. doi:10.1371/journal.pone.0172864.
Graham, H., Kauffman, R. & Khaliq, W. (2022) Colorectal Cancer Screening Prevalence, Perceived Barriers, and Preference for Screening Colonoscopy Among Hospitalized Women. The Turkish Journal of Gastroenterology. 33 (11), 901–908. doi:10.5152/tjg.2022.21567.
Haggar, F. A., Boushey, R. P. (2009). Colorectal Cancer Epidemiology: Incidence, Mortality, Survival, and Risk Factors. Clinics in Colon and Rectal Surgery
Havranek, R. (2013) What is an Open Access Colonoscopy? Russell Havranek, MD. https://russellhavranekmd.com/open-access-colonoscopy/.
Hewitson, P., Glasziou, P., Irwig, L., Towler, B. & Watson, E. (2007) Screening for colorectal cancer using the faecal occult blood test, Hemoccult. The Cochrane Database of Systematic Reviews. 2007 (1), CD001216. doi:10.1002/14651858.CD001216.pub2.
Hossain, Md.S., Karuniawati, H., Jairoun, A.A., Urbi, Z., Ooi, D.J., John, A., Lim, Y.C., Kibria, K.M.K., Mohiuddin, A.K.M., Ming, L.C., Goh, K.W. & Hadi, M.A. (2022) Colorectal Cancer: A Review of Carcinogenesis, Global Epidemiology, Current Challenges, Risk Factors, Preventive and Treatment Strategies. Cancers. 14 (7), 1732. doi:10.3390/cancers14071732.
Hull, M.A., Rees, C.J., Sharp, L. & Koo, S. (2020) A risk-stratified approach to colorectal cancer prevention and diagnosis. Nature Reviews Gastroenterology & Hepatology. 17 (12), 773–780. doi:10.1038/s41575-020-00368-3.
Hull, T. & Church, J.M. (1994) Colonoscopy–how difficult, how painful? Surgical Endoscopy. 8 (7), 784–787. doi:10.1007/BF00593441.
Janout, V., Kollárová, H. (2001). Epidemiology of colorectal cancer. Olomouc: Biomedical papers of the Medical Faculty of the University Palacky. https://doi.org/10.5507/bp.2001.00
Johnson, I. T., & Lund, E. K. (2007). Review article: nutrition, obesity and colorectal cancer. Alimentary pharmacology & therapeutics. https://doi.org/10.1111/j.1365-2036.2007.03371.x
Klabunde, C.N., Lanier, D., Breslau, E.S., Zapka, J.G., Fletcher, R.H., Ransohoff, D.F. & Winawer, S.J. (2007) Improving colorectal cancer screening in primary care practice: innovative strategies and future directions. Journal of General Internal Medicine. 22 (8), 1195–1205. doi:10.1007/s11606-007-0231-3.
Lansdorp-Vogelaar, I., Knudsen, A. & Brenner, H. (2010) Cost-effectiveness of colorectal cancer screening – an overview. Best practice & research. Clinical gastroenterology. 24 (4), 439–449. doi:10.1016/j.bpg.2010.04.004.
Lewandowska, A., Rudzki, G., Lewandowski. T., Stryjkowska-Góra, A., Rudzki, S. (2022). Risk Factors for the Diagnosis of Colorectal Cancer. Jaroslaw: SAGE Publications https://doi.org/10.1177/10732748211056692
Lynch, H. T., Lynch, J. F., Lynch, P. M., Attard, T. (2008). Hereditary colorectal cancer syndromes: molecular genetics, genetic counseling, diagnosis and management. Familial cancer https://doi.org/10.1007/s10689-007-9165-5
Ma, X., Zhang, B. & Zheng, W. (2014) Genetic variants associated with colorectal-cancer risk: comprehensive research synopsis, meta-analysis, and epidemiological evidence. Gut. 63 (2), 326–336. doi:10.1136/gutjnl-2012-304121.
McMichael, A. J., McCall, M. G., Hartshorne, J. M., Woodings, T. L. (1980). Patterns of gastrointestinal cancer in European migrants to Australia: the role of dietary change. International journal of cancer. https://doi.org/10.1002/ijc.2910250402.
MD, N.D. & MD, L.R. (2022) Colon cancer screening decisions: What’s the best option and when? 15 June 2022. Harvard Health. https://www.health.harvard.edu/blog/colon-cancer-screening-decisions-whats-the-best-option-and-when-202206152762 [Accessed: 29 July 2023].
National Institutes of Health. (2006). What You Need To Know About Cancer of the Colon and Rectum. Bethesda, MD: U.S. Department of Health and Human Services & National Institutes of Health
Navarro, M., Nicolas, A., Ferrandez, A. & Lanas, A. (2017) Colorectal cancer population screening programs worldwide in 2016: An update. World Journal of Gastroenterology. 23 (20), 3632–3642. doi:10.3748/wjg.v23.i20.3632.
Park, C.H., Jung, Y.S., Kim, N.H., Lee, M.Y., Park, J.H., Park, D.I. & Sohn, C.I. (2019) Impact of temperature and humidity on performance of the fecal immunochemical test for advanced colorectal neoplasia. Scientific Reports. 9, 9824. doi:10.1038/s41598-019-44490-y.
Qaseem, A., Crandall, C.J., Mustafa, R.A., Hicks, L.A., Wilt, T.J., & for the Clinical Guidelines Committee of the American College of Physicians* (2019) Screening for Colorectal Cancer in Asymptomatic Average-Risk Adults: A Guidance Statement From the American College of Physicians. Annals of Internal Medicine. 171 (9), 643–654. doi:10.7326/M19-0642.
Rawla, P., Sunkara, T. & Barsouk, A. (2019) Epidemiology of colorectal cancer: incidence, mortality, survival, and risk factors. Przegla̜d Gastroenterologiczny. 14 (2), 89–103. doi:10.5114/pg.2018.81072.
Song, L.-L. & Li, Y.-M. (2016) Current noninvasive tests for colorectal cancer screening: An overview of colorectal cancer screening tests. World Journal of Gastrointestinal Oncology. 8 (11), 793–800. doi:10.4251/wjgo.v8.i11.793.
Stauffer, C.M. & Pfeifer, C. (2023) Colonoscopy. In: StatPearls. Treasure Island (FL), StatPearls Publishing. p. http://www.ncbi.nlm.nih.gov/books/NBK559274/.
Theresa (2022) Risk Stratification 101: What is it and how is it used? 5 July 2022. Johns Hopkins ACG® System. https://www.hopkinsacg.org/risk-stratification-101-what-is-it-and-how-is-it-used/ [Accessed: 29 July 2023].
World Cancer Research Fund and American Institute for Cancer Research. (2007). Food, Nutrition, Physical Activity, and the Prevention of Cancer: A Global Perspective. Washington, DC: American Institute for Cancer Research
Zheng, S., Schrijvers, J.J.A., Greuter, M.J.W., Kats-Ugurlu, G., Lu, W. & de Bock, G.H. (2023) Effectiveness of Colorectal Cancer (CRC) Screening on All-Cause and CRC-Specific Mortality Reduction: A Systematic Review and Meta-Analysis. Cancers. 15 (7), 1948. doi:10.3390/cancers15071948
Zisman, A. L., Nickolov, A., Brand, R. E., Gorchow, A., Roy, H. K. (2006). Associations between the age at diagnosis and location of colorectal cancer and the use of alcohol and tobacco: implications for screening. Archives of internal medicine. https://doi.org/10.1001/archinte.166.6.629




