Supervised by: Yuhui Zhou, BA (Hons). Yuhui is a 5th year medical student at the University of Cambridge. She gained a First class degree in her intercalated year studying Pathology. She has an interest in Cancer & Immunology and has been awarded a Wellcome Trust Biomedical Vacation Scholarship to study host responses to infection.
Abstract
Background:
The glioblastoma multiforme is one of the most aggressive brain tumours occurring in the central nervous system. Despite many the treatments that exist for cancer, invasive, surgical and other, none was found to be effective enough to cure significant amounts of cases of glioblastoma. Patient outcomes remain poor, with less than 25% making it to two years and fewer than 10% making it to five. Two major factors contributing to poor survival rates is the inefficient drug delivery across the blood-brain barrier and the tumour cells’ development of drug resistance. Due to gliomas being caused by accumulation of genetic alterations in glial cells, gene therapy has shown promising results for future treatment as it overcomes certain limitations of conventional therapy. Viral vectors have been studied in numerous clinical trials but have yet to reach FDA approval, having complications with delivery, high cytotoxicity and insufficient tumour penetration. Non-viral vectors are currently being evaluated in clinical studies and are showing great promise as potential delivery vehicles in treatment of the glioblastoma.
Objective:
In this literature review, we will compare viral and non-viral vector delivery systems and their efficiency in treating the glioblastoma multiforme.
Methods:
Google Scholar and PubMed databases, as well as numerous journals including MDPI, BMJ and NCBI, were searched for research articles and literature reviews from 2011 to present, as older studies had different results and were not as developed. The variables included cytotoxicity, efficiency when combined with other treatments, efficiency in penetrating the blood-brain barrier, risk of developing resistance to the treatment and survival rate.
Results:
Most reviews came to the conclusion that even though viral vector gene therapy is more developed than non-viral gene therapy, it still shows promise for future treatments. Viral vectors, initially having shown great promise, were revealed to develop drug resistance after a while, therefore being a temporary solution. Liposomal were proven to be effective in inducing apoptosis when accompanied by another treatment, otherwise having temporary gene expression. Polymers were revealed to be rather effective in increasing survival due to their small size and receptiveness to chemical modifications. These modifications are very important as without them, polymers can be relatively toxic. And lastly, RNA nanoparticles were capable of reaching intracranial tumours and reducing the oncogene’s expression.
Introduction
The glioblastoma, also known as the glioblastoma multiforme, is the most frequent and aggressive primary malignant tumour of the central nervous system. It occurs in the brain and spinal cord, more often in older adults. These tumours are typically located in troublesome areas of the cerebral hemisphere and have an extremely high rate of infiltration into surrounding tissue, consequently having a poor prognosis. Without therapy, patients die within three months. And patients treated with optimal therapy, such as radiation therapy or chemotherapy, have an approximate survival of twelve months, with fewer than 25% making it to two years and approximately 5% making it to five (Kwiatkowska et al., 2013; Caffery, Lee, Alexander-Bryant, 2019).
A variety of treatments with the use of drugs such as temozolomide (TMZ), bis-chloronitrosourea (BCNU) and cisplatin which interfere with cell growth and therefore prevent tumour growth, were used in patients. However, their delivery’s efficiency was limited due to chemotherapeutic resistance and the Blood-Brain Barrier, which is a cell barrier that regulates ionic concentrations to allow synaptic signaling, while also regulating transport to the brain and protecting it from neurotoxins by preventing the entry of certain cells and molecules (Caffery, Lee, Alexander-Bryant, 2019).
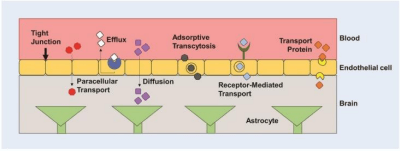
Figure 1: Methods of transporting molecules across the blood-brain barrier. Paracellular transport, diffusion, adsorptive transcytosis, receptor-mediated transport, transport protein (Caffery, Lee, Alexander-Bryant, 2019).
The reason why gene therapy was proposed as a potential working treatment is because gliomas are caused by an accumulation of genetic mutations in glial cells. However, it is less straightforward in the case of cancer, as tumours develop and continue to grow through these accumulated abnormalities. So, in order for this to work, the notion of using gene therapy as treatment for cancer had to be broadened to include delivery of therapeutic genes to the tumour, resulting in the death of cancerous cells or the enhancement of immune responses against them. Researchers have thus been working on different methods to come up with viral and non-viral techniques of delivering treatment effectively and achieving a therapeutic effect. Viruses used to be the typical genetic material carriers, but alternative vehicles such as liposomes, polymers and RNA-based nanoparticles have also been extensively studied and developed, and are currently being evaluated in clinical trials. The purpose of this review is to describe these approaches and compare their advantages and issues.
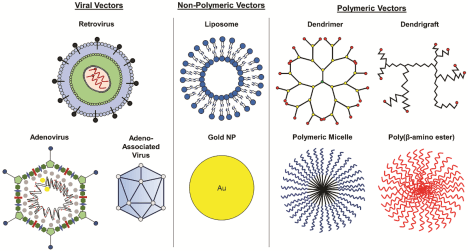
Figure 2: Various vectors used in gene therapy for treating GBM (Caffery, Lee, Alexander-Bryant, 2019).
Methods
The researching methods we have used when producing this scientific article review are the following. When looking for sources, all of us were considering the trustworthiness, relevance of the articles to our research topic and currency – the publish date of the articles. Our research was done primarily with the help of search engines such as Google Scholar and sources considered were mostly scientific journals such as BMJ, MDPI and NCBI. After choosing a specific topic, we have searched for as much information on it as possible. Most of the articles that were used to create this review article were first, review articles by which we gained an overview of what was done in the field so far and then experiment research articles that had a specific question that they were answering and were more focused on finding a concrete result.
We found that older articles, ranging from 1995 to 2010, showed different results and conclusions than the newer ones, which all had rather similar results, starting from 2011, until present.
The strengths of using this method of gathering information is the accessibility to big amounts of information on the internet. The articles considered as sources for this article are also written by the top experts in the field which supports accuracy and reliability. The limitations, on the other hand, could be the review articles being subject to biases, influenced by the author’s viewpoints etc. Further limitations could also be potential errors in the transfer of data from primary sources to our review article. Omission of other relevant research to the topic could lead to incomplete conclusions and results.
Results
Viral Vectors
Viral vectors were the first delivery vehicles used in gene therapy for treating gliomas. The most common viral vectors that are currently being used for GBM treatment in clinical trials are neurotrophic retroviruses and adenoviruses. These viruses have the capacity to integrate into the host chromosomes, which in turn will allow their DNA to permanently stay in the host cells, and result in long-lasting expression of the gene of interest (Lundstrom, Boulikas, 2003). However, recent studies have proven that eventually, the tumour cells will develop resistance to these vectors.
2.1. Adenovirus
Adenoviruses are non-enveloped, double-stranded DNA viruses. Such vectors have an advantage over other viral vectors – they have a low level of toxicity and are capable of carrying large DNA molecules, and thus have been thoroughly studied for GBM gene therapy. A good example is the tumour-suppressing Ad-p53 construct, combining the Ad5 vector, which introduces an intact complementary DNA copy of the p53 gene and the p53 protein. This combination restores the wild-type p53 function, which induces apoptosis (Caffery, Lee, Alexander-Bryant, 2019).
It was proven to successfully transfect astrocytic cells, with minimal toxicity, when intratumourally injected. The cells were however, only detected within 5mm of the injection site, meaning that this adenoviral vector’s ability to penetrate the tumour is rather limited, therefore will not have an extremely significant effect (Nandi, Lesniak, 2019). Presence of a host antibody to the adenovirus was also discovered, meaning that the tumour cells developed resistance to the treatment (Chen et al., 2014).
2.2. Retrovirus
The best studied retrovirus and suicide gene is the human herpes simplex virus (HSV) derived enzyme – thymidine kinase (HSV-tk). This enzyme catalyses the phosphorylation of cytotoxic nucleoside analogues that can be incorporated into the DNA of proliferating cells, thus preventing DNA replication and halting cell division (Caffery, Lee, Alexander-Bryant, 2019). Moreover, this enzyme has to be used with a prodrug called ganciclovir. It converts the drug into its active form, ganciclovir triphosphate, which will then inhibit DNA replication and cell division in HSV-tk transfected cells, as mentioned above. However, the results of this study showed that HSV-tk transfection was limited and therefore would only be effective in treating smaller tumours.
Despite the initial success and promise of the aforementioned studies in suicide gene therapy for glioma, it was later proven that even HSV-tk cells could develop resistance to the prodrugs, thus needing combination with chemo-radiotherapy (Caffery, Lee, Alexander-Bryant, 2019).
Non-Viral Vectors
3.1. Liposomes
Liposomes are artificial, lipid-based microvesicles used to deliver drugs and peptides into cells, however, recent studies have shown that chemical engineering of lipids will allow the formation of stable DNA-lipid associations that can be used as a gene delivery vehicle (Kwiatkowska et al., 2013).
SGT-53 is a complex containing a cationic liposome and a human wild type p53 DNA sequence carried by a plasmid. This complex has been proven to successfully and efficiently deliver therapeutic genetic material, containing the p53 sequence, to the tumour, thus crossing the blood-brain barrier. However, it was shown to be most effective in enhancing apoptosis in combination with another agent, the temozolomide (TMZ) prodrug. Combining the two treatments inhibited tumour growth, increased apoptosis and significantly prolonged survival. In contrast, using TMZ alone did not have any effect on median survival and the tumour cells even developed resistance to the drug. Consequently, combining TMZ and SGT-53 limits development of resistance to the prodrug, therefore making this treatment more effective (Kim, et al., 2015).
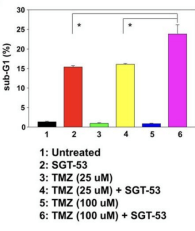
Figure 3: Diagram depicting effectiveness of treatment in fractions of DNA (Ref. Chang, 2014).
3.2. Polymers
Cationic polymers are macromolecules that bind DNA using electrostatic interactions. They are often used in cell transfection as they offer many advantages, such as small size and flexibility that permit modifications, which will in turn improve tumour targeting and distribution (Kwiatkowska et al., 2013).
Polyethylenimine (PEI) is a well-known example of a linear polymer used in delivery of plasmids. It has the capacity to strongly bind DNA, as well as being highly efficient in transfection. However, it would need further modifications in order to reach intracranial tumours, and without said modifications, it could be highly toxic (Pulkkanen, Yla-Herttuala, 2005). By adding a functional group like poly-ethileneglycol (PEG), PEI has shown significant and permanent improvements in circulation and the tumour stroma. Furthermore, after having added myristic acid to PEI polymers, they were able to cross the blood-brain barrier, therefore successfully delivering plasmids to intracranial gliomas and prolonging survival.
3.3. RNA Nanoparticles
Spherical nucleic acids (SNAs) are interference-based RNA nanoparticles that are able to penetrate the cerebral hemisphere. They are composed of gold nanoparticle cores and densely packed small interfering RNA (siRNA) oligonucleotides.
During the first trial conducted on humans, patients with GBM were treated with Nu-0129 (siBcl2L12-SNAs), administered intravenously, followed by tumour resection. Results revealed that there were no treatment-related toxicities. Furthermore, evaluation of the resected tumours depicted that intravenously administered SNAs did in fact, reach intracranial tumours. The drug’s delivery into the tumour cells resulted in significant reduction of the Bcl2L12 protein expression. SNA nanoparticles could therefore be a promising potential approach to treating GBM (Kumthekar et al., 2021).
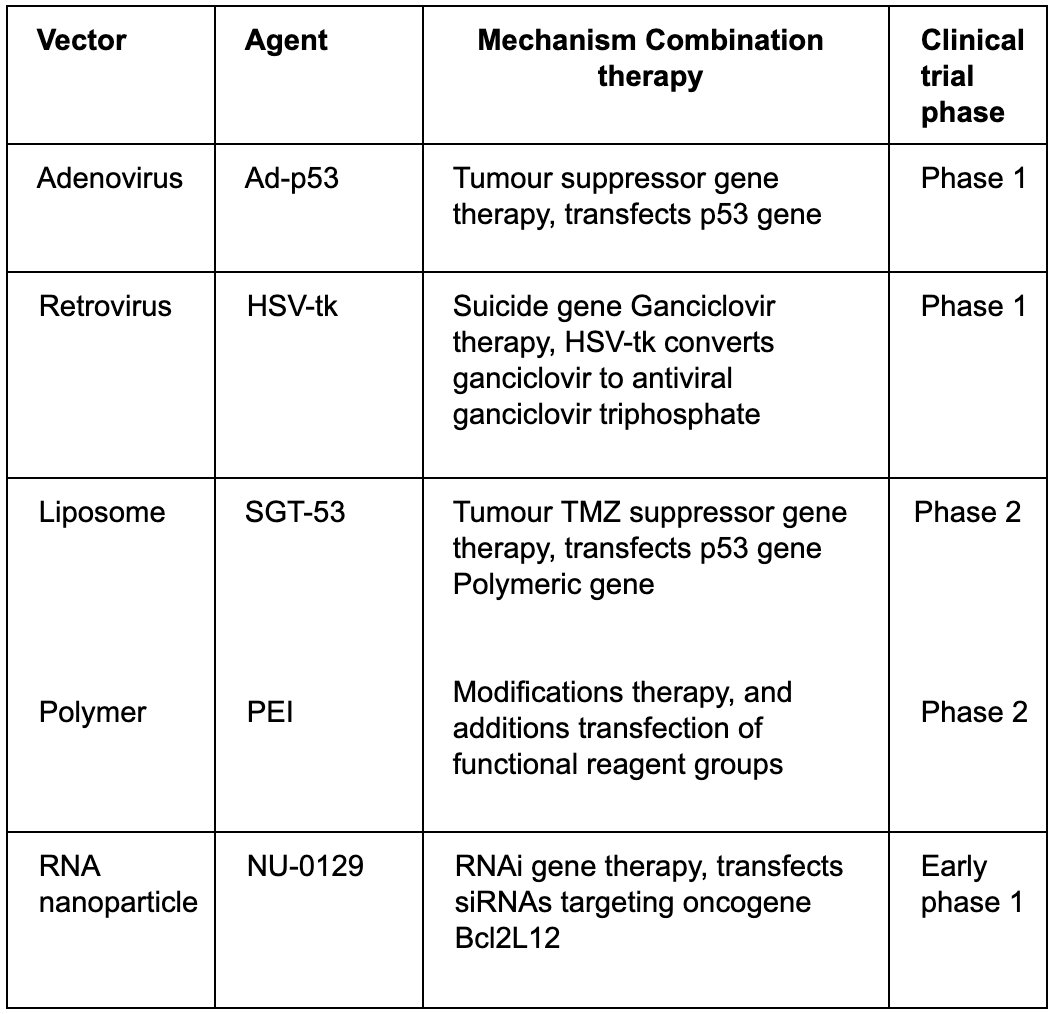
Table 1: Examples of viral and non-viral vectors used in treating GBM, as well as the agents being used, a brief description of their mechanism, whether or not they require additional treatment, and the stage of their clinical trial (Caffery, Lee, Alexander-Bryant, 2019; Salameh et al., 2019).
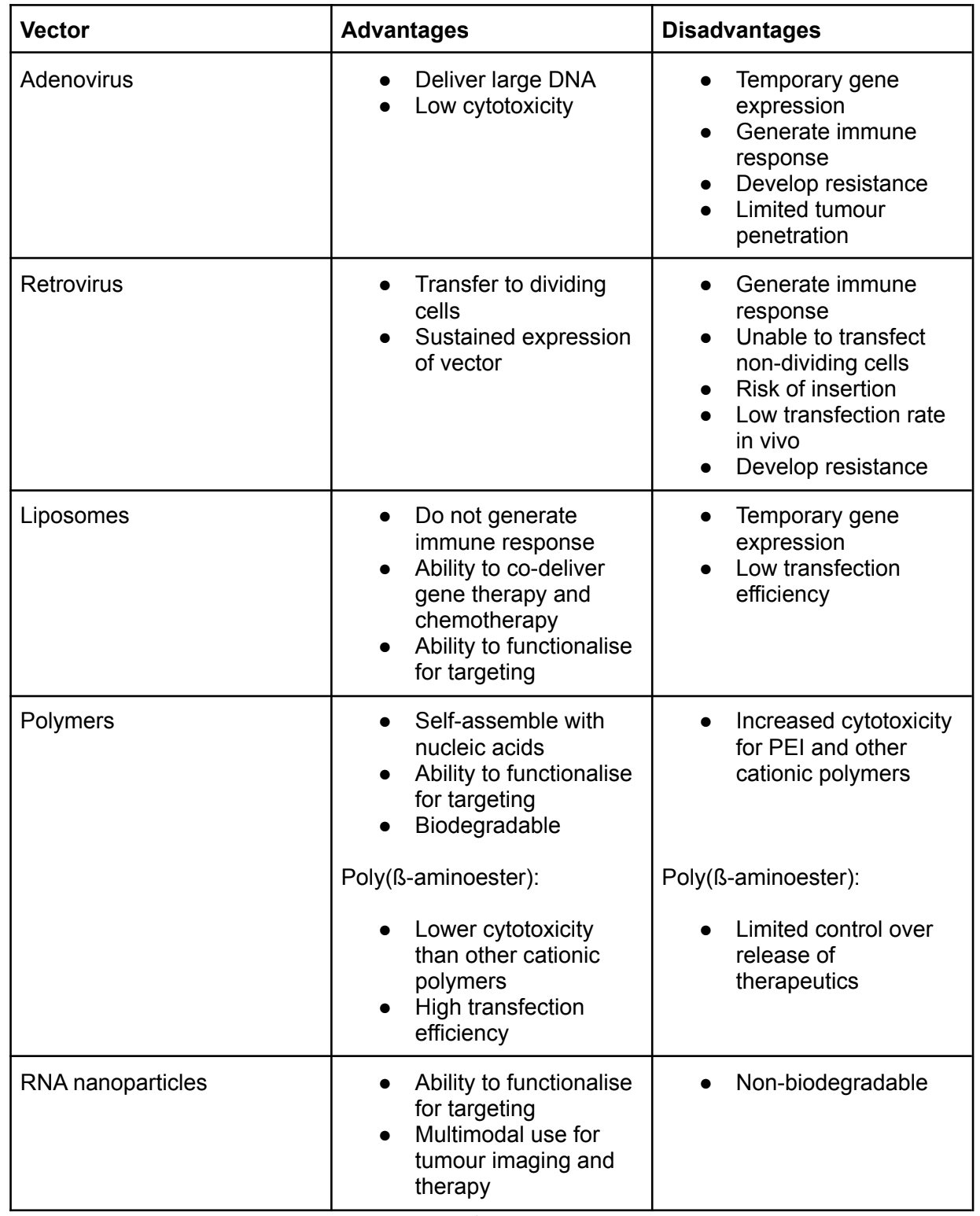
Table 2: The advantages and disadvantages of certain viral and non-viral vectors (Salameh et al., 2019; Tzeng et al., 2011).
Discussion
In this review, viral and non-viral vectors that have been used in glioblastoma gene therapy are discussed. Additionally, the differences, advantages and disadvantages of the two methods are demonstrated.
Whilst viral vectors have proven to be rather efficient in transfecting cells, safety concerns regarding their use in humans are clear, making non-viral vectors an attractive alternative.
On one hand, adenoviral vectors are able to carry DNA molecules of impressive size while retroviral vectors are capable of transferring into dividing cells, while also being able to retain their gene expression. On the other hand, adenoviral vectors are only able to express their genes temporarily and retroviral vectors are lacking the ability of transfecting non-dividing cells, which in turn, is responsible for their low transfection efficiency. Both types of viral vectors were also proven to generate an immune response, meaning that after a while, tumour cells would get accustomed to the treatment and the therapy would no longer have a significant effect. Lastly, both methods were ineffective after a while due to the tumour cells developing resistance.
In contrast, all non-viral vectors discussed in this review are able to functionalise to fit their target, which means that they are able to undergo numerous chemically engineered modifications and additions to their structure in order to be able to properly inhibit further proliferation and induce apoptosis.
Liposomal vectors are very advantageous because they do not generate an immune response from the organism, however the same cannot be said for polymers and RNA nanoparticles, which are capable of causing inflammatory cytokines, as well as having an impact on the liver. It was concluded that they did not result in significant immunoreactions, although the selection of safe gene sequences should still be considered an important factor. Much like viral vector approaches, liposomes have low transfection abilities and temporary gene expression. We should also highlight that most non-viral vectors work better when combined with other treatments, or modifications. Different types of polymeral vectors have their own advantages and disadvantages, all of them are however, biodegradable and can self-assemble with nucleic acids. The linear polymer that we focused on in this review, the PEI, was proven to be relatively cytotoxic. Nevertheless, a good alternative was found – the poly(ß-aminoester), which has a lower level of cytotoxicity and is highly efficient in transfection. Lastly, we looked at RNA nanoparticles, specifically the Nu-0129, which has a low level of cytoxicity, and was shown to reduce the oncogene’s expression and proved useful as a marker in tumour imaging.
To summarise everything that has been said, the main issue with using viral vectors to treat GBM, is that the tumour cells will eventually develop resistance to the treatment, which is a tremendous disadvantage. However, non-viral vectors still lack necessary development and research in certain areas, although they are definitely a great alternative.
Bibliography
- Caffery B, Lee J, Alexander-Bryant A. Vectors for Glioblastoma Gene Therapy: Viral & Non-Viral Delivery Strategies. Nanomaterials. 2019;9(1):105. doi:10.3390/nano9010105
- Lundstrom K, Boulikas T. Viral and non-viral vectors in gene therapy: technology development and clinical trials. Technology in Cancer Research & Treatment. 2003;2(5):471-486. doi:10.1177/153303460300200513
- Chen G, Zhang S, He X, Liu S, Ma C, Zou XP. Clinical utility of recombinant adenoviral human p53 gene therapy: current perspectives. OncoTargets and therapy. 2014;7(3099067):1901-1909. doi:10.2147/OTT.S50483
- Nandi S, Lesniak MS. Adenoviral virotherapy for malignant brain tumors. Expert Opinion on Biological Therapy. 2009;9(6):737-747. doi:10.1517/14712590902988451
- Kim SS, Rait A, Kim E, Pirollo KF, Chang EH. A tumor-targeting p53 nanodelivery system limits chemoresistance to temozolomide prolonging survival in a mouse model of glioblastoma multiforme. Nanomedicine: Nanotechnology, Biology and Medicine. 2015;11(2):301-311. doi:10.1016/j.nano.2014.09.005
- Research Gate. ResearchGate | share and discover research. ResearchGate. Published 2019. https://www.researchgate.net/
- Kwiatkowska A, Nandhu M, Behera P, Chiocca E, Viapiano M. Strategies in Gene Therapy for Glioblastoma. Cancers. 2013;5(4):1271-1305. doi:10.3390/cancers5041271
- Kumthekar P, Ko CH, Paunesku T, et al. A first-in-human phase 0 clinical study of RNA interference–based spherical nucleic acids in patients with recurrent glioblastoma. Science Translational Medicine. 2021;13(584):eabb3945. doi:10.1126/scitranslmed.abb3945
- Pulkkanen KJ, Yla-Herttuala S. Gene Therapy for Malignant Glioma: Current Clinical Status. Molecular Therapy. 2005;12(4):585-598. doi:10.1016/j.ymthe.2005.07.357
- National Cancer Institute. Using Oncolytic Viruses to Treat Cancer. National Cancer Institute. Published February 9, 2018. https://www.cancer.gov/news-events/cancer-currents-blog/2018/oncolytic-viruses-to-treat -cancer
- Boado RJ. RNA interference and nonviral targeted gene therapy of experimental brain cancer. NeuroRX. 2005;2(1):139-150. doi:10.1602/neurorx.2.1.139
- Tzeng SY, Guerrero-Cázares H, Martinez EE, Sunshine JC, Quiñones-Hinojosa A, Green JJ. Non-viral gene delivery nanoparticles based on Poly(β-amino esters) for treatment of glioblastoma. Biomaterials. 2011;32(23):5402-5410. doi:10.1016/j.biomaterials.2011.04.016
- Salameh JW, Zhou L, Ward SM, Santa Chalarca CF, Emrick T, Figueiredo ML. Polymer‐mediated gene therapy: Recent advances and merging of delivery techniques. WIREs Nanomedicine and Nanobiotechnology. 2019;12(2). doi:10.1002/wnan.1598




