Supervised by: Amanda Liu, BEng, MSc. Amanda spent her undergraduate years studying Biochemical Engineering at UCL (University College London), where she was awarded First Class Honours. She then completed her Master’s degree in Clinical and Therapeutic Neuroscience at the University of Oxford. She is currently studying Medicine (Graduate Entry) at the University of Cambridge.
Januvia
Januvia (generic name: sitagliptin) is a type of prescribed inhibitor that is used to treat adult patients living with type 2 diabetes. Taken as an oral diabetes tablet dail,y along with a balanced diet and exercise, can increase insulin and reduce glucagon in the body. The aim of the inhibitor is to reduce the blood sugar levels (glucose), thus minimising the risks associated with type 2 diabetes.
Discovered by Merck & Co. Inc., the Januvia inhibitor is the result of continuous optimisation of a potent and selective triazolopiperazine series. As written by Ann E. Webber and Nancy Thornberry, the glucagon-like peptide 1 (GLP-1), was seen as a valid treatment for type 2 diabetes and dipeptidyl peptidase 4 (DPP-4) was used as an alternative method of GLP-1 treatment. Both led to an inhibitor programme from Merck in 1999. Multiple DPP-4 inhibitors were used to begin with, but were eventually dropped due to toxicity in rat and dog safety studies. It was hypothesised that this was because the inhibitors used would inhibit the other proline peptidases DPP-8 and DPP-9, resulting in profound toxicity. This then led to the development of a highly selective DPP-4 inhibitor that would only affect the desired enzymes, thus eventually culminating in the optimisation of two screening hits, giving a highly selective beta-amino acid piperazine series, where further refinement resulted in the identification of sitagliptin, or Januvia.
As for how Januvia affects the body, it is essential to note that this prescription is only used to treat type 2 diabetes and is not used to aid with type 1 diabetes or diabetic ketoacidosis (increased ketones in blood and urine). By adjusting the insulin and glucose the body makes after eating, blood sugar levels can be kept at a normal level. In doing so, it tries to remove the two major issues with type 2 diabetes: a lack of insulin produced in the pancreas and an overproduction of glucose in the liver. An intake of 100mg at the same time each day before or after a meal is the standard dosage, and should a diet and exercise be used alongside the prescription, control of blood sugar levels is greatly improved. Januvia works by reducing the rate at which the incretin hormones (GLP-1 and GIP) are deactivated. These incretin hormones are part of the system that usually ensures blood sugar levels are at an acceptable level and are released by the intestine during the day and after a meal. Usually, they rapidly lose activity due to the enzyme dipeptidyl peptidase 4 (DPP-4). However, Januvia is part of the DPP-4 inhibitor class and will block the deactivation of the incretin hormones, thus increasing the length of time of active incretin levels. This in turn increases the amount of insulin released by the pancreas while reducing glucagon levels and glucose produced from the liver, which aids significantly in treating type 2 diabetes.
As is usual with prescribed drugs, the Januvia inhibitor does have specific side effects when taken into the body. Headaches accompanied by other aches and pains are among the most common effects experienced by patients. Nausea and bloating have also been found to be a common side effect, resulting in patients needing to avoid rich or spicy foods. Furthermore, nose and throat infections along with swollen feet or ankles can also occur when Januvia is taken. Finally, Januvia has been found to cause severe stomach (abdominal) pain in several patients; this could mean the patient has pancreatitis (inflamed pancreas) and should consult a doctor immediately. It is clear that Januvia, while still essential in dealing with type 2 diabetes, is not exempt from harming patients and should be taken with care, ensuring that diet and exercise accompanies the daily intake of 100mg.
SGLT2
SGLT2 inhibitors are a class of medications that modulate sodium-glucose transport proteins in the nephron. SGLT2 inhibitors have shown unprecedented benefits for those with type 2 diabetes; they help to lower the blood sugar in those with type 2 diabetes. SGLT2 medications include canagliflozin, dapagliflozin and empagliflozin. The SGLT2 inhibitors work by preventing the kidneys from reabsorbing sugar back into the bloodstream. This allows blood sugar levels to remain moderately low, allowing excess glucose to be removed via urine.
They are FDA-approved for use with diet and exercise to lower blood sugar in adults with type 2 diabetes. Medicines in the SGLT2 inhibitor class include canagliflozin, dapagliflozin, and empagliflozin. They are available as single-ingredient products and also in combination with other diabetes medicines such as metformin. SGLT2 inhibitors lower blood sugar by causing the kidneys to remove sugar from the body through the urine. The safety and efficacy of SGLT2 inhibitors have not been established in patients with type 1 diabetes, and FDA has not approved them for use in these patients.
Although SGLT2 inhibitors are effective at slowing the progression of kidney disease, reducing heart failure, and lowering the risk of kidney failure and death in people with kidney disease, type 2 diabetes can lead to severe problems, including blindness, nerve and kidney damage, and heart disease.
Comparison
In this passage, the price and effectiveness are the two factors used to compare the two main types of inhibitor therapy for type 2 diabetes. Insulin and Synjardy are another two groups we used for comparison in order to compare the overall inhibitors group and other common therapies.
Effectiveness is measured through the reeducation in A1C. The A1C test—also known as the hemoglobin A1C or HbA1c test—measures average blood sugar levels over the past 3 months. According to research conducted by the FDA, insulin has the highest reduction in A1C levels.
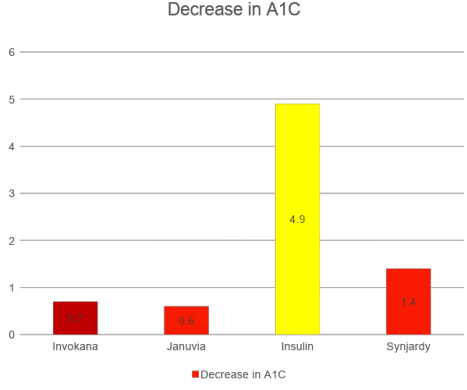
The graph above shows that the inhibitor drugs have the smallest decrease in A1C levels. Compared to Invokana and Januvia, Synjardy and insulin significantly reduce the A1C level.
Beyond effectiveness, cost is another crucial factor that must be considered. It directly impacts which therapy is accessible to any given patient. Here, the data from CVS Pharmacy, an American retail corporation, is used to compare the retail prices of these drugs on the American market.
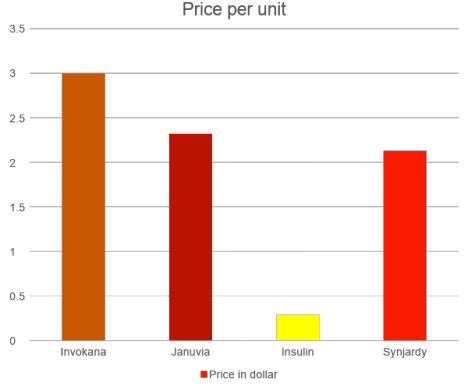
The graph clearly shows that insulin is the cheapest, despite the insulin price increasing significantly as a result of the shortage due to the COVID-19 pandemic. Synjardy has almost the same price as Januvia. Invokana is the most expensive of those drugs.
Even with health insurance, people in the U.S. find it difficult to afford inhibitor therapy. People who cannot afford these options tend to purchase insulin as a substitute. However, improper use of insulin could cause serious side effects including confusion and loss of consciousness. The high price of these inhibitors drugs can therefore lead to more severe damage for patients.
Although their prices are high, the inhibitor therapies do have a unique advantage. They act when patients need them to and will not when patients do not need it. Unlike other drugs, the inhibitors do not significantly impact the mechanism of the human body. If prices can be lowered in the future, these medications will become the major therapies for type-2 diabetes.
Reference:
Center for Drug Evaluation and Research. (n.d.). Sodium-glucose cotransporter-2 (SGLT2) inhibitors. U.S. Food and Drug Administration. Retrieved September 4, 2022, from https://www.fda.gov/drugs/postmarket-drug-safety-information-patients-and-providers/so dium-glucose-cotransporter-2-sglt2-inhibitors
Januvia, inn: Sitagliptin – european medicines agency. (n.d.). Retrieved September 5, 2022, from https://www.ema.europa.eu/en/documents/scientific-discussion/januvia-epar-scientific-dis cussion_en.pdf
Liu, J., Zhou, H., Zhang, Y., Fang, W., Yang, Y., Huang, Y., & Zhang, L. (2020, May 11). Reporting of immune checkpoint inhibitor therapy–associated diabetes, 2015–2019. American Diabetes Association. Retrieved September 4, 2022, from https://doi.org/10.2337/dc20-0459
UpToDate. (n.d.). Retrieved September 4, 2022, from https://www.uptodate.com/contents/sodium-glucose-co-transporter-2-inhibitors-for-the-tr eatment-of-hyperglycemia-in-type-2-diabetes-mellitus




