Neurodiversity and Personality
Neurodiversity recognises that human brains naturally vary and that traits associated with Attention Deficit Hyperactivity Disorder (ADHD) and Autism Spectrum Disorder (ASD) represent part of typical neurological variation rather than inherent pathology (Cheney, n.d; De Domenico et al., 2025). Due to natural variation, individuals with ADHD frequently exhibit impulsivity, distractibility and difficulty sustaining attention, while autistic individuals may have challenges with social communication and interpreting social cues (Schriber, Robins & Solomon, 2014). From this perspective, differences in attention, emotional regulation and social behaviour are viewed as natural variations in cognition rather than deficits. The expression of these traits varies across contexts and life experiences, making them more nuanced than simple “strengths” or “weaknesses”, making their behaviour more nuanced than simply labelling traits as good or bad (Cheney, n.d.; Schriber, Robins & Solomon, 2014).
Individuals with ADHD may struggle to maintain focus, switch between tasks or control immediate reactions, which may affect academic performance and interpersonal functioning (Martinez et al., 2026). In contrast, autistic individuals often portray a diverse perspective on social engagement, as shown through difficulty in interpreting social cues such as sarcasm, understanding emotional expressions such as common idioms or phrases, or initiating social interaction, often leading to misunderstandings with neurotypical individuals (Schriber, Robins & Solomon, 2014).
Neurodiverse individuals’ emotional regulation further shapes how neurodivergent traits are expressed in everyday functioning (De Domenico et al., 2025). These emotional tendencies interact with attention and social behaviour, creating complex, individualised profiles. Understanding these interactions helps distinguish relatively stable personality traits from context-dependent behaviours, reducing the risk of misinterpreting neurodivergent traits as pathological (Schriber et al., 2014), which is crucial to avoid misinterpreting neurodivergent traits as pathological behaviours (Schriber, Robins & Solomon, 2014).
Adolescents and adults with ADHD often resemble traits better associated with disinhibition, such as impulsivity, distractibility and risk-taking, compared to their neurotypical peers (De Domenico et al., 2025). On the other hand, autistic individuals tend to have higher neuroticism, a personality trait characterised by a greater tendency to experience negative emotions such as anxiety, sadness, irritability or emotional instability (Costa & McCrae, 1992), and lower extraversion, agreeableness, conscientiousness and openness, suggesting that neurodivergence shapes how traits are expressed (Schriber, Robins & Solomon, 2014). Conceptualising personality traits as dimensions rather than categories allows researchers to study these differences without automatically pathologising them (De Domenico et al., 2025; Schriber, Robins & Solomon, 2014).
Understanding the intersection between neurodivergence and personality is particularly important during adolescence and young adulthood, the periods marked by ongoing emotional and identity development. Examining these traits within a dimensional, neurodiversity-informed framework may clarify how neurodivergent characteristics interact with personality patterns to shape social and emotional outcomes.
This study examines the hypothesis that neurodivergent adolescents and young adults will demonstrate higher neuroticism and disinhibition, alongside lower conscientiousness and extraversion. By taking a dimensional and informed perspective on how neurodiverse traits develop, how they interact with neurodivergent characteristics, and how they influence social and emotional outcomes without reducing individuals to labels or stereotypes, insightful understanding can be achieved (De Domenico et al., 2025; Schriber, Robins & Solomon, 2014).
OCEAN 5 Traits and Neurodivergency Relation Through Experiences
Findings about the relationship between the Five Factor Model (FFM) – a psychological framework categorising human personality into five dimensions – and neurodivergency suggest that fluency in personality dimensions can offer a valuable framework for understanding experiential differences between neurotypical and neurodivergent individuals.
Similarly, adolescents with ADHD were found to have significantly higher levels of disinhibition. including increased impulsivity, distractibility, irresponsibility and risk-taking using the DSM-5 Alternative Model of Personality. Given the FFM framework, this pattern corresponds strongly to lower conscientiousness and aspects of higher neuroticism (a tendency to experience negative emotions) (De Domencio et al., 2024).
These patterns reflect individual differences with ADHD who tend to score lower on conscientiousness, displaying an increased difficulty mediating relationships between executive function and inattention (Krieger et al., 2020). Higher neuroticism in ASD individuals is associated with increased emotional reactivity, anxiety and distress, highlighting how environmental disruptions may be experienced more intensely. Lower extraversion within individuals reflects reduced social motivation or comfort in highly stimulating interpersonal contexts, while lower agreeableness relates to differences in social reciprocity rather than hostility (Schribler et al., 2014). Reduced conscientiousness has been linked to differences in executive functioning, while lower openness to experience may reflect a preference for consistency and order in structured environments, rather than diminished creativity (Schriber, Robins & Solomon, 2014). Neurodivergent distress may therefore arise less from trait levels themselves and more from environments that demand high extraversion, conscientiousness and rapid cognitive flexibility. High neuroticism in ASD may partially reflect chronic sensory overstimulation and low extraversion may be secondary to sensory load in social environments. Together, they highlight the need to enhance assessment and treatment for autistic individuals.
The FFM is useful here because it describes stable tendencies in emotional reactivity, social orientation, self-regulation and cognitive flexibility – domains that closely overlap with common challenges reported by autistic and ADHD populations. Rather than treating neurodivergence as a single category of impairment, trait-based profiles can clarify how and where mismatches with typical school and social environments are most likely to occur. Overall, the FFM helps explain why autistic distress may be amplified in environments that demand rapid social adaptation, constant novelty and sustained self-management (Schriber, Robins & Solomon, 2014).
Recognising that dispositional variations are part of human diversity itself promotes more flexible, inclusive practices that support lifelong learning for all students, regardless of their diagnostic status. These trait-environment mismatches have important implications beyond clinical settings, particularly in education (Nicolosi & Dillenburger, 2025). Traditionally, autism has been viewed through a medical model that categorises individuals based on observed behaviours, resulting in support needs that vary widely across different people. The neurodiversity movement (NDM) challenges this perspective, framing autism as a natural variation in human neurology rather than a disorder to be cured. It advocates for societal acceptance, inclusion and environmental adaptation, emphasising autism as an intrinsic aspect of identity rather than a deficit to be corrected. This shift encourages education systems to move away from diagnosis-driven interventions and instead focus on accommodating diverse ways of learning and participating. Debates surrounding interventions such as Applied Behaviour Analysis (ABA) highlight tensions between supporting skill development and respecting individual autonomy. Proponents argue that personalised, evidence-based approaches can enhance quality of life, independence and social participation (Nicolosi & Dillenburger, 2025), while critics raise concerns about potential impacts on identity and wellbeing.
Research into neurodiversity and education emphasises that traditional, diagnosis-centred models often create divisions amongst students. These diagnosis models marginalise those who do not meet specific criteria. Increasingly, neurodiversity is recognised as a broad, dimensional spectrum, acknowledging that most individuals possess some neurodivergent traits. Recognising the spectrum of neurodiversity allows for more inclusive educational practices that accommodate a wide range of developmental and cognitive differences, rather than focusing solely on diagnosed conditions. Fixed labels such as “neurotypical” can be limiting and deterministic, often overlooking the fluid and varied nature of neurological differences resulting from personality differences (Fisher, 2024a). As a result, there is a growing realisation that education needs to be more flexible and personalised, moving away from rigid, standardised systems that tend to blame or pathologise children who do not fit the norm. Emphasising understanding and accommodating individual differences, particularly in areas such as executive functioning and emotional regulation, is seen as essential. Self-directed education models may better accommodate sensory regulation and autonomy needs among neurodivergent learners, from the perspective of the FFM. Overall, embracing unique personalities within all individuals calls for a shift towards more inclusive, flexible and child-centred educational practices that better support the diverse ways in which children develop and learn, regardless of any prior diagnosis (Fisher, 2024).
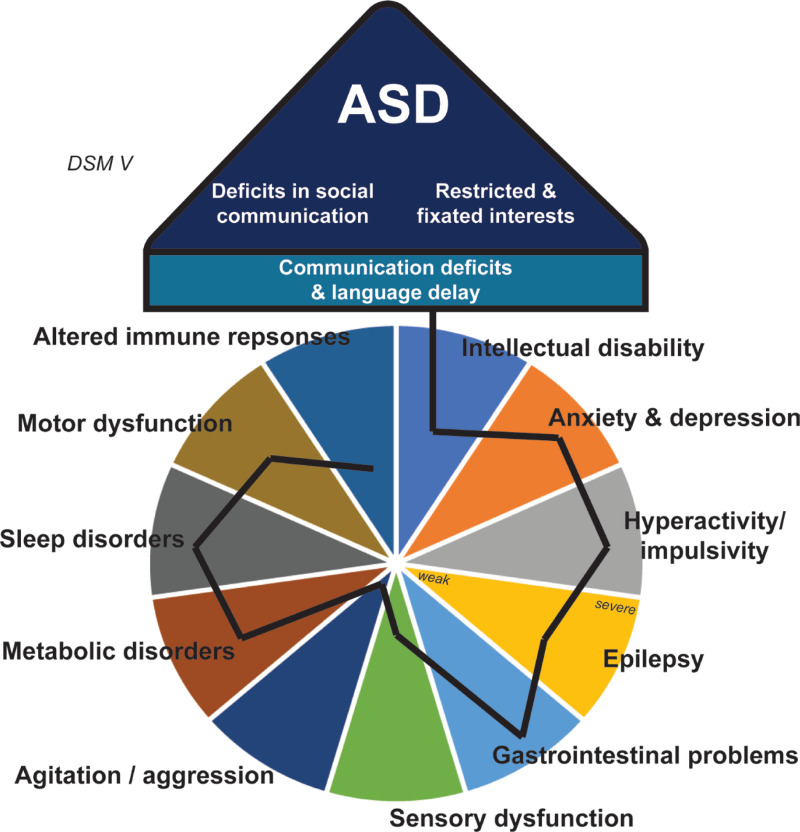
Figure 1. Spectrum of neurodivergent traits and conditions, emphasising that individuals may have a mix of these traits in varying degrees. The image supports the idea that neurodiversity comprises a wide range of differences and promotes a shift toward more flexible, inclusive approaches in education and society that recognise individual variability. Adapted from Sauer et al. (2021).
Perspectives on Neurodivergence and Personality Disorders
An individual’s personality develops from experiences, environment and inherited factors and remains relatively stable over time. Personality disorders are mental health conditions characterised by deeply ingrained, inflexible patterns of thinking, feeling and behaving that deviate markedly from cultural expectations. These patterns typically emerge in adolescence or early adulthood and are evident across situations, causing significant distress or functional impairment (American Psychiatric Association, 2022). When such traits become extreme and rigid, interfering with personal relationships and daily life, criteria for a personality disorder may be met. The DSM-5-TR groups personality disorders into three clusters (A, B and C), reflecting patterns of eccentric, dramatic or anxious behaviour (American Psychiatric Association, 2022).
Although personality disorders and neurodevelopmental conditions are conceptually distinct, their behavioural features can overlap in clinically meaningful ways, often resulting in misdiagnosis, diagnostic overshadowing and inappropriate treatment. Shared features such as emotional dysregulation, impulsivity, social difficulties and relationship challenges contribute to the confusion (Dell’Osso et al., 2023; Hours et al., 2022). For example, autistic adults frequently exhibit higher neuroticism and lower extraversion, agreeableness, conscientiousness and openness than neurotypical peers (Schriber et al., 2014). ADHD is associated with elevated disinhibition and risk-taking, corresponding to lower conscientiousness and higher neuroticism (Ditrich et al., 2021). A key difference is that neurodevelopmental conditions such as ASD and ADHD are present from early life, whereas personality disorders generally emerge later (American Psychiatric Association, 2022; World Health Organisation, 2019). Shared features such as emotional dysregulation, impulsivity and social or relational difficulties contribute to diagnostic ambiguity, despite differing underlying mechanisms.
Misdiagnosis is attributed to substantial symptom overlap between ASD, ADHD and personality disorders, as attention problems, impulsivity and social difficulties may appear similar across conditions despite differing aetiologies (Kentrou et al., 2024; Iversen & Kildahl, 2022; Dell’Osso et al., 2023). Features such as attention problems, impulsivity and social challenges appear in all three, although causes differ: sensory processing differences in ASD, executive functioning deficits in ADHD and relational stressors (e.g., fears of abandonment) in Borderline Personality Disorder (BPD). Misdiagnosis is particularly common among women and girls, in part due to masking of autistic traits (Dell’Osso et al., 2023; Hull et al., 2017). High rates of ASD-ADHD co-occurrence further complicate differential diagnosis (Hours et al., 2022). Accurate differentiation is clinically essential as it enables targeted interventions and improves outcomes (World Health Organisation, 2019).
Recognising these distinctions is necessary, as many symptoms of BPD – such as emotional instability and impulsivity – overlap with those seen in ADHD and ASD, making careful assessment critical for providing appropriate and effective support (Kenézlői et al., 2025).
Borderline personality disorder involves the following symptoms: intense emotions, rapid mood swings and low self-esteem. BPD is also often linked to self-injurious behaviours. Individuals with BPD frequently fear abandonment, which leads to “black-and-white thinking”, where they swing between idealising and devaluing others. These patterns typically develop during adolescence, influenced by temperament and environmental factors (American Psychiatric Association, 2022). Many people with BPD report childhood trauma, such as emotional, physical or sexual abuse, which contributes to emotional dysregulation and attachment issues (Zanarini et al., 2012; Weiner et al., 2019).
Genetically, BPD shows moderate heritability involving vulnerabilities in: the serotonin system, which affects impulse control and emotional reactivity; the dopamine system, linked to stress-related reward dysregulation; and the HPA axis, indicating heightened stress responses (Ditrich et al., 2021). While no single gene causes BPD, a family history increases the risk. Neurobiological mechanisms, such as fronto-limbic dysregulation and serotonin-dopamine interactions, overlap with traits seen in neurodevelopmental disorders like ADHD and ASD (Ditrich et al., 2021; Dell’Osso et al., 2023). Masking behaviours can complicate diagnosis and contribute to secondary borderline traits (Hull et al., 2017; Dell’Osso et al., 2023). The onset differs: ASD is usually identified early in childhood, whereas BPD often emerges in adolescence (American Psychiatric Association, 2022; World Health Organisation, 2019).
BPD frequently co-occurs with ADHD, sharing features such as impulsivity, emotional instability and interpersonal challenges. Impulsivity in BPD tends to worsen under stress (Rüfenacht et al., 2019; Ditrich et al., 2021). Treating ADHD symptoms may improve emotional regulation and reduce impulsivity, supporting integrated treatment approaches (Rüfenacht et al., 2019). Overall, BPD results from a complex interplay of biological, psychological and environmental factors. Understanding its overlap with ASD and ADHD is essential for accurate diagnosis and effective, personalised treatment (Ditrich et al., 2021; Weiner et al., 2019).
Beyond BPD, many neurodivergent individuals, including those with ADHD, ASD and tic disorders, face overlapping behavioural and emotional challenges that can make daily life overwhelming. Individuals with ASD often stim intentionally because it helps them regulate sensory overload. When overwhelmed with information, emotional overload may lead to exhaustion. Social interactions can also be exhausting, particularly when individuals engage in masking behaviours (Hull et al., 2017).
Anxiety and Obsessive Compulsive Disorder (OCD) are also related to ASD. Anxiety may stem from sensory overload and difficulty interpreting social cues, which differs from social anxiety rooted in fear of negative evaluation. Executive function differences further contribute to attentional difficulties (Ditrich et al., 2021). ASD, ADHD and tic disorders are related in complex ways and require a nuanced understanding.
In conclusion, personality disorders such as BPD and neurodivergent conditions, including ADHD and ASD, are deeply interconnected, sharing challenges in emotional regulation, impulsivity and executive functioning (Ditrich et al., 2021; Dell’Osso et al., 2023; Weiner et al., 2019). Individuals living with these conditions often face sensory overload, masking, stimming, compulsive behaviours and attentional difficulties, which can make daily life demanding and socially complex (Hull et al., 2017). Recognising both shared features and individual experiences supports a neurodiversity-informed approach to assessment and intervention, enabling more precise, compassionate and effective support.
Neurodiversity in the Developing Brain
ASD is a neurodevelopmental condition associated with differences in brain development, cognition and behavioural presentation (Lord et al., 2020; American Psychiatric Association, 2013). Accumulating evidence suggests that ASD originates from premature differences in brain development, questioning views that primarily attribute it to environmental factors (American Psychiatric Association, 2013). Contemporary neuroscience increasingly frames ASD through the lens of neurodiversity, emphasising natural variation in neural development rather than deficit-focused models. This shift has sparked evolving research into the biological mechanisms behind atypical brain maturation, particularly in synaptic organisation and neural connectivity.
Synaptogenesis is one of the most essential processes in childhood brain development. Synaptogenesis entails the formation of synapses between neurons, which are connections that underpin neural communication. This surge in synaptic density provides the structural substrate for neuroplasticity and experience-dependent learning (Huttenlocher & Dabholkar, 1997). This is followed by either synaptic pruning or selective elimination of excess synapses. Synaptic pruning occurs to refine neural circuits and enhances cognitive efficiency as well as executive functioning (Lord et al., 2020). The process of synaptic pruning is conventional during typical neural development. These successive developmental waves are essential for establishing excitatory-inhibitory (E/I) signalling and coherent large-scale networks (Huttenlocher & Dabholkar, 1997). Emerging findings suggest that some autistic individuals exhibit dissimilarity in synaptic pruning, which may lead to irregular patterns in information processing, indicating cognitive variability (Tang et al., 2014). Furthermore, advances in developmental neuroscience and genetics have highlighted the role of gene regulation to shape synaptic structures (Lord et al., 2020; Zoghi & Bear, 2012). Such findings support theories that ASD is not simply a behavioural condition, but one that is found in variation about neurobiological processes.
Synaptic pruning alone is unlikely to be solely responsible for shaping cognitive profiles (Lord et al., 2020), as longitudinal studies display atypical patterns of brain growth in autistic children, including early brain overgrowth and altered cerebral cortex development, which take place during critical periods (Courchesne et al., 2011). Post-mortem cortical analyses have demonstrated increased dendritic spine density in autistic individuals, attributed to reduced or delayed synaptic pruning during development (Tang et al., 2014). The formation of neural networks involved in executive functioning, focus and learning is influenced by this deviation, which may in turn be responsible for the extent of cognitive performance (Lord et al., 2020).
Although accumulating evidence implicates early neurodevelopmental mechanisms in ASD, the extent to which synaptogenesis and synaptic pruning directly determine cognitive ability remains contested. The body of knowledge surrounding the neurobiology of ASD continues to grow; however, existing literature predominates on behavioural characteristics, leaving a gap in findings that explain the role of early neurodevelopmental processes and synaptic pruning in ASD’s cognitive potential. Thus, this portion of the paper seeks to answer the following: to what extent do early neurodevelopmental processes and synaptic pruning contribute to cognitive ability in ASD?
Synaptic Pruning and Cognitive Development in Neurodiverse Conditions
AUTISM SPECTRUM DISORDER AND PRUNING DYSREGULATION
Synaptic pruning is a key mechanism of cortical development, involving activity-dependent elimination of redundant synapses to refine neural circuits, in ASD (a neurodevelopmental condition that is characterised by variations in cognition and behaviour) (American Psychiatric Association, 2013; Lord et al., 2020). Atypical pruning may contribute to the altered cortical organisation present in ASD. Normative post-mortem analyses demonstrate peak synaptic density in early childhood followed by progressive reduction in the number of synapses through adolescence, particularly in the prefrontal cortex (PFC) (Huttenlocher & Dabholkar, 1997). Therefore, deviations from this pattern could influence executive functioning and cognitive efficiency.
Evidence from ASD brain tissue examination indicates an elevated dendritic spine density in cortical projection neurons (Hustler & Zhang, 2010), suggesting delayed or reduced pruning in autistic individuals. However, the small sample size and cross-sectional design of the study limit its generalisability. In addition, while some neuroimaging studies report similar patterns of early brain overgrowth, others show differences where regions or the extent of growth are affected (Courchesne et al., 2011; Ecker et al., 2015). These findings indicate that whilst microstructural pruning deficits translate to observable macrostructural patterns and cognitive outcomes, these outcomes and patterns can vary across neurodiverse populations. Moreover, it is revealed that the timing of pruning, rather than how much pruning, may better shape cognitive outcomes in ASD.
mTOR signalling regulates protein synthesis and autophagy – both important components for synaptic turnover. Tang et al (2014) found that hyperactive mTOR in mouse models disrupts autophagy, leading to excess synapses and resembling ASD behaviours. While informative, translational caution is required as mouse models cannot fully capture human cognition. Additional genetic studies further support the idea that synaptic regulation is disrupted in ASD, with many associated variants affecting synaptic scaffolding and chromatin modification (Bourgeon, 2015). Although rare de novo mutations can have strong effects, most risk arises from polygenic backgrounds, suggesting that pruning is not a result of a single gene.
Therefore, anatomical, genetic and mechanistic evidence suggests that disrupted synaptic pruning in ASD, specifically altered developmental timing, rather than an excess of synapses, is a more likely explanation for cognitive differences between neurotypical and autistic individuals.
ADHD AND SYNAPTIC PRUNING
The principle that existing ASD findings suggest (that disrupted synaptic pruning associates with altered cortical organisation and reduction of synapses, particularly in the prefrontal cortex, affecting executive function) widely relates to other neuropsychiatric disorders, including ADHD, demonstrating that the delay in such processes ultimately leads to hyperactivity, affected behaviour and cognitive functioning. Disturbed synaptic pruning has been linked to disruptions in neural circuitry that contribute to behavioural difficulties, and genetic variations affecting proteins involved in synapse formation, development, plasticity and pruning have been associated with several neuropsychiatric disorders, including ASD and ADHD (Eltokhi et al., 2020).
Microglia, the brain’s immune cells, make up about 10-15% of cells in the central nervous system and are vital to this process because they remove underused synaptic connections during development (Eltokhi et al., 2020). Moreover, synaptic refinement involves dendritic spines, which are dynamic protrusions that form the primary sites of excitatory synapses and change in density, shape and function throughout foetal and postnatal development in response to activity (Ugarte et al., 2023). Dysregulated pruning can cause abnormal increases or decreases in spine density, a pattern suggested to reinforce the neurodevelopmental basis of disorders such as ADHD. Therefore, disrupted pruning can lead to grey matter changes specific to each disorder, with distinct structural profiles reflecting unique pruning effects (Eltokhi et al., 2020). In ADHD, neuroimaging studies have shown subtle but significant, non-progressive reductions in grey and white matter, particularly in cortico-striatal regions that mature later in adolescence.
Clinically, ADHD is characterised by persistent inattention, hyperactivity and impulsivity that affect behaviour and cognitive functioning. It often occurs with ASD, sharing genetic influences and challenges in executive functioning, sensory processing and social communication (Eltokhi et al., 2020). Evidence implies that ADHD in childhood may reflect delayed brain maturation due to slower synaptic pruning, especially in the prefrontal cortex, which supports inhibitory control, attention and working memory, helping explain why many individuals’ symptoms improve as they reach adulthood (Eltokhi et al., 2020). Together, these findings suggest that dysregulated synaptic pruning and dendritic spine development play an essential role in the structural and functional brain differences seen in ADHD and many other neuropsychiatric disorders.
Collectively, evidence from ADHD suggests that alterations in synaptic pruning and cortical maturation do not disrupt cognition globally, but instead exert regionally specific effects depending on the developmental trajectory of neural systems (Shaw et al., 2007; Petanjek et al., 2011). Consequently, variations in the spatial disruption and timing of synaptic refinement may give rise to distinct cognitive phenotypes, a principle that becomes especially salient in dyslexia, where language-related cortical networks are selectively affected (Pugh et al., 2000; Richlan, Kronbichler & Wimmer, 2009).
DYSLEXIA AND ATYPICAL CORTICAL CONNECTIVITY
Evidence from ASD and ADHD suggests that alterations in synaptic pruning and cortical maturation exert regionally specific effects depending on the developmental trajectory of particular neural systems (Shaw et al., 2007; Petanjek et al., 2011). Dyslexia further exemplifies this principle, demonstrating how a disruption in refinement within language-dominant cortical networks meticulously impairs phonological processing and reading (Plugh et al., 2000; Richlan, Kronbichler & Wimmer, 2009).
Comparative findings from ASD provide a critical framework for understanding how early synaptic refinement influences cognitive outcomes in dyslexia. The overlapping language phenotypes observed in ASD and dyslexia have led researchers to propose shared genetic and neurodevelopmental contributors (Eicher & Gruen, 2014; Eicher et al., 2015). Rather than representing isolated pathologies, converging genetic and neuroimaging evidence suggests that both conditions may arise from disruptions in early synaptic organisation that subsequently shape cognitive function and cortical connectivity (Frith, 2013).
Dyslexia is characterised by chronic and pronounced impairments in reading that are not attributed to deficits in intelligence or cognitive ability (Çetin et al., 2025). Neuroimaging studies consistently demonstrate altered structural and functional connectivity in the left temporo-parietal and occipito-temporal regions, which are essential for phonological decoding and fluency in reading (Richlan, Kronbichler & Wimmer, 2014; see Figure 2).
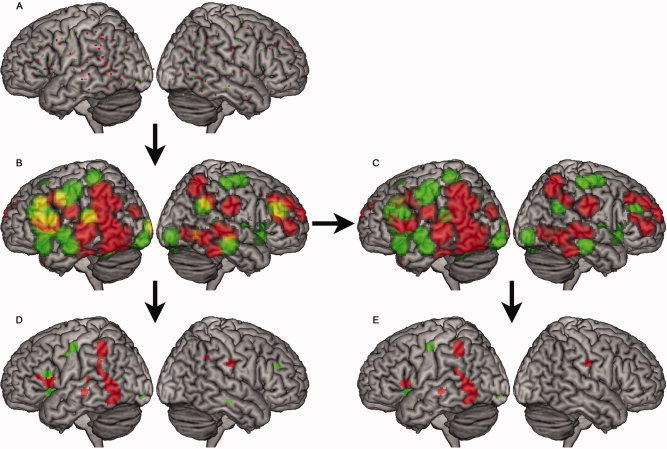
Figure 2. Meta-analytic activation differences between individuals with dyslexia and atypical readers during reading tasks, highlighting reduced activation in left temporo-parietal and occipito-temporal regions. Adapted from Richlan, Kronbichler & Wimmer (2014).
Similarly, ASD is typically associated with atypical patterns of cortical connectivity, displaying an increase in local connectivity alongside reduced long-range integration. Such deviations likely result from disrupted activity-dependent synaptic pruning during key developmental periods.
Synaptogenesis during early childhood results in an overproduction of synaptic connections, followed by activity-dependent microglia-mediated pruning to refine and augment network efficiency. Microglia play a central role in this polishing process, with complement proteins including C1q and C3 tagging redundant synapses for elimination, enabling precise refinement of functional circuits (Çetin et al., 2025). Dysregulation of this procedure, whether through excessive synaptic elimination or insufficient pruning, alters the equilibrium between local and long-range cortical connectivity. Such imbalances are implicated in neurodevelopmental conditions, including ASD, where atypical connectivity patterns are often observed, showing increased local connectivity alongside reduced long-range integration (Frith, 2013). In language-related cortical regions, such disruptions impair the fine-tuning of phonological processing networks, leading to the characteristic reading deficits observed in dyslexia. In ASD, altered pruning has been hypothesised to contribute to atypical network synchronisation and language-related impairments, further supporting a shared mechanistic pathway.
The frequent co-occurrence of dyslexia with ASD-related traits and other neurodevelopmental problems reinforces the notion that early deviations in synaptic refinement may produce broad neurocognitive vulnerability rather than isolated deficits (Brimo et al., 2021). Thus, atypical synaptic pruning provides a plausible developmental mechanism by which shared genetic risk factors shape overlapping yet distinct cognitive profiles.
Early Neurodevelopmental Processes and Cognitive Outcomes in Neurodiverse Conditions
ATYPICAL GROWTH TRAJECTORIES AND SYNAPTIC REFINEMENT IN ASD
Early atypical neurodevelopmental trajectories indicate divergence (from trajectories of neurotypical individuals) during critical periods of circuit formation in ASD patients. Longitudinal MRI studies report accelerated total brain volume growth between 6 and 24 months in children later diagnosed with ASD, particularly in frontal and temporal regions associated with executive and social cognition (Courchesne et al., 2011). Enlargement is not consistently present at birth, however, implying altered postnatal growth dynamics rather than congenital structural abnormalities (American Psychiatric Association, 2013; Lord et al., 2020).
However, replication of findings in similar studies has been variable, suggesting early brain overgrowth is not universal (Courchesne et al., 2011; Ecker et al., 2015). Regional structural analyses show differences in cortical thickness and white matter organisation, though findings vary across age and methodology (Ecker et al., 2015). Functional connectivity studies suggest increased local connectivity with altered long-range integration (Uddin et al., 2014), which may influence executive function and social processing. Limitations include motion artefacts and cohort heterogeneity, emphasising the need for cautious interpretation.
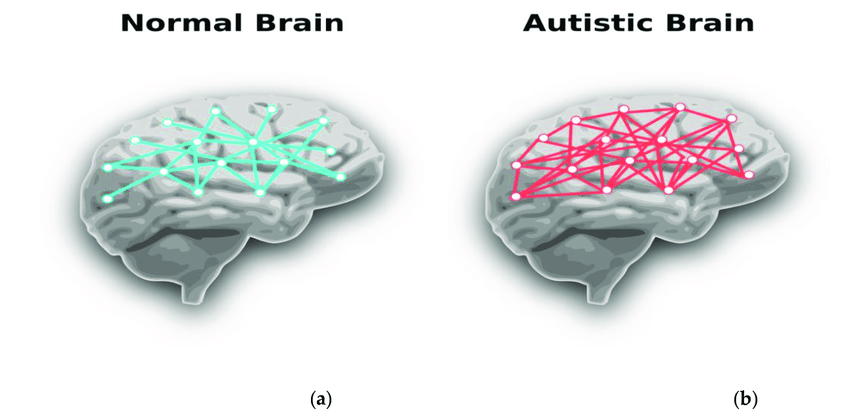
Figure 3. Conceptual illustration of differences in local versus long-range connectivity in autism spectrum conditions, showing atypical connection patterns relative to typical neural networks. Adapted from Baura et al. (2022).
Genetic evidence intersects with imaging data, highlighting genetic variants associated with increased risk of ASD that affect synaptic plasticity and circuits maturation (Bourgeon, 2015). Multiple studies, all pointing to this same conclusion, suggest that early growth divergence may reflect knock-on effects of disrupted activity-dependent processes (like synaptic pruning) rather than deterministic outcomes. The probabilistic nature of polygenic influence also implies heterogeneity in developmental outcomes.
Overall, converging imaging and genetic evidence support a developmental systems model in which altered timing of growth and synaptic refinement shapes network organisation in ASD. This all demonstrates that different cognitive profiles in autistic individuals emerge from dynamic divergence in neurodevelopmental trajectories rather than static structural abnormalities, reinforcing the importance of early neural processes in understanding ASD-related cognition. This concept of early neurodevelopment shaping cognitive outcomes extends to other neurodiverse conditions, such as ADHD, where similar mechanisms influence developmental trajectories and cognitive profiles.
DELAYED CORTICAL MATURATION AND COGNITIVE OUTCOMES IN ADHD
Early neurodevelopmental processes are essential in shaping cognitive outcomes in children, particularly in neurodiverse conditions such as ADHD. Disruptions in key brain regions, such as the prefrontal cortex and cerebellum, are associated with ADHD symptoms. The cerebellum is crucial for procedural learning, affecting attention, motor control and executive functions (Arnett et al., 2013). In ADHD, atypical cerebellar development is linked to cognitive deficits like impaired attention and impulse control (Betz et al., 2015). Children with ADHD also show delays in cortical maturation, especially in the PFC and basal ganglia, regions involved in working memory and response inhibition (Arnett et al., 2013). These developmental delays contribute to persistent cognitive impairments in ADHD, emphasising the importance of early neurodevelopmental disruptions. Early-life psychosocial deprivation, such as institutional rearing, increases the risk of ADHD, suggesting environmental factors also play a role (Arnett et al., 2013).
In ADHD, early neurodevelopmental disruptions, particularly in the cerebellum and PFC, lead to cognitive impairments. The cerebellum supports procedural learning, which is crucial for attention regulation and executive functions. Disruptions in cerebellar development impair these functions, contributing to ADHD symptoms like attention difficulties and impulse control (Liu et al., 2020). ADHD is also associated with delays in cortical development, particularly in the PFC and basal ganglia regions essential for cognitive functions such as attention and memory (Betz et al., 2015). Children with ADHD often experience delays of 2-5 years in cortical development, which can contribute to their ongoing cognitive and behavioural challenges (Betz et al., 2015). Furthermore, early-life deprivation such as environmental factors influence neurodevelopment in ADHD (Arnett et al., 2013).
SHARED LANGUAGE PHENOTYPES AND GENETIC OVERLAP (ASD & DYSLEXIA)
Contrasting to the parallel impact of environmental factors on both ASD and ADHD, a genetic link bridges the gap between ASD and dyslexia. Comparable language phenotypes of ASD and dyslexia lead to the assumption that the two conditions branch from the same “genetic contributors” (Eicher & Gruen, 2014). Researchers Eicher and Gruen (2014) found links between dyslexia and ASD through the following risk genes: ATP2C2 and MRPL19 (language impairment and dyslexia, respectively). Due to these genes, autistic and dyslexic children struggle to achieve language fluency, a core developmental milestone in neurotypical children (Eicher & Gruen, 2014). Symptoms of early language impairment include a lack of “phonological processing” and “verbal language” (Eicher & Gruen, 2014).
In terms of reading, a study conducted by Frith (2013) revealed that children in Italy could read the Italian language easily, suggesting that the Italian language may be an optimal environment for dyslexic children, and the issue may be in the syntax and appearance of common letters and words. Despite this phenomenon, Frith’s (2013) study reveals that underlying neurocognitive processes remain in all dyslexic and neurodiverse individuals.
A number of both autistic and dyslexic individuals demonstrate compensation, a behaviour that allows these individuals to easily pass as typical in “standard diagnostic tests” (Frith, 2013). In an experimental treatment option, Johnson et al.’s (2021) study reveals that dyslexic children responded significantly more to ADHD medications than other neurodiverse patients, although they began with lower baseline scores.
Discussion
This paper examined how neurodiversity, brain growth and personality relate, noting a move from old ways of seeing conditions like ADHD and ASD as flaws to a more accepting, varied view. In the past, neurodivergent traits were primarily seen as medical problems, concentrating on what people were unable to do. Yet, the neurodiversity paradigm can be seen as reframing these features as biological variations in human neurobiology instead of pathologies. Personal characteristics (e.g., impulsivity, attentional variability and social communication differences) are progressively seen as part of normal neurological variety, which may influence how help and interventions are assessed. This view encourages consideration of whether traditional deficit-focused models might overlook individual strengths.
At the neurobiological level, early developmental processes such as synaptogenesis and synaptic pruning play a central role in shaping cognitive and behavioural outputs. Synaptogenesis refers to the formation of neural connections during childhood, followed by activity-dependent pruning that refines these networks. Unusual pruning patterns, including delayed or reduced pruning, have been linked to ASD and ADHD. For example, expanded dendritic spine density in ASD suggests reduced pruning, potentially contributing to differences in social cognition and information processing. In ADHD, postponed cortical development, particularly in the prefrontal cortex, is linked to difficulties in executive functioning and impulse regulation. These findings highlight the importance of developmental timing and regional specificity in brain maturation, implying that variations can have noteworthy influences on behaviour and could interact with environmental needs.
Genetic and neuroimaging research additionally reveals that neurodiversity comes from complex polygenic influences affecting synaptic plasticity and neural connectivity. Variations in pathways like mTOR are likely to alter developmental trajectories, contributing to the heterogeneity observed across neurodevelopmental profiles. Patterns of advanced regional connectivity alongside fewer long-range integrations have been identified in ASD, imaging differences in information processing networks, which may help present some behavioural differences.
Taking on a dimensional perspective also clarifies links between neurodivergent traits and personality. Autistic individuals often display higher neuroticism and lower extraversion, while ADHD traits are associated with impulsivity and lower conscientiousness. Recognising these traits along a continuum may reduce stigma and support more personalised interventions. Overall, integrating neurodevelopmental and personality frameworks promotes a strengths-based understanding of neurodiversity and highlights the importance of considering individual context, which could encourage inclusive approaches that support wellbeing across an individual’s lifespan.
Another important point that emerged throughout this paper is the intersection between neurodiverse personality traits and personality disorders. While on one hand, personality disorders typically emerge in adolescence or early adulthood, they should not be diagnosed earlier due to hormonal and cognitive development, and are characterised by pervasive, inflexible and maladaptive patterns. ASD and ADHD, on the other hand, are neurodevelopmental conditions with onset in early childhood. Understanding the overlap of these neurobiological systems and disorders is crucial for correct treatment and therapeutic outcomes.
Neurodevelopmental differences forego character appearance, whereas personality disorder traits evolve stably and context-insensitively over time. Personality disorder traits require stability across contexts, while neurodivergent traits often fluctuate depending on environmental factors.
Symptoms, for example, between borderline personality disorder and ADHD often overlap, complicating diagnosis and treatment. Emotional regulation, impulse control and social cognition may appear unstable across disorders. Evidence in this paper suggests ADHD is associated with higher disinhibition and lower conscientiousness, BPD with high neuroticism and impulsivity, and ASD with higher neuroticism and lower extraversion, though individual variation exists. Mentioned traits are best conceptualised using dimensional personality models (e.g., Big Five), allowing overlap without collapsing distinct diagnoses. Trait peak does not necessarily imply a personality disorder.
Further research indicates that individuals with personality disorders often have histories influenced by childhood trauma, attachment disruption and chronic relational stress. In contrast, neurodevelopmental conditions are influenced more strongly by polygenic risk and early neural development. Personality disorders show medium heritability for traits like emotional dysregulation, while ASD, for example, shows significant familial aggregation.
In addition, personality disorder features arise when network-level regulation fails to stabilise, while neurodivergent traits mirror enduring, context-sensitive disparities. Diagnostic overshadowing and masking, especially in autistic females, further complicate diagnosis.
These differences underline the importance of looking at each person’s experiences and environment. Neurodivergent traits can change over time, unlike the more stable patterns seen in personality disorders, so support needs to be flexible and adaptable. Tracking how these traits unfold can help improve care and understanding. Given the necessity for research to remain current, future studies should focus on network-level regulation, prefrontal-amygdala interactions and longitudinal neurodevelopment to define when patterns qualify as clinical disorders and to inform developmentally-sensitive interventions.
Conclusion
Neurodivergence emerges not just as discrete pathology but rather as a dynamic variation in the development of the brain and personality expression. Covering ASD, ADHD and dyslexia, emerging evidence suggests that differences in synaptogenesis, cortical maturation and network connectivity shape cognitive profiles. Across reflecting global impairments, conditions similar to these continue to arise from altered developmental timing and region-specific patterns of neural organisation.
More importantly, these neurobiological patterns and variations continue to intersect with measurable personality dimensions. Traits such as neuroticism, conscientiousness, disinhibition and extroversion simply do not exist independently of neurodevelopment; instead, they reflect how underlying cognitive systems regulate attention, emotion and social behaviour. When viewed dimensionally, neurodivergence and its characteristics align with broader patterns of human variability rather than categorical deficit. Distress often arises not solely from trait differences themselves, but environments structured around narrow expectations of flexibility, attention and social interaction.
Distinguishing neurodevelopmental conditions from personality disorders remains essential clinically, particularly given overlapping behavioural features. Although shared mechanisms such as emotional regulation challenges or executive function differences necessitate more precise, compassionate and individualised approaches to support.
Ultimately, the integration of neuroscience with personal theory strengthens a neurodiverse informed framework. Cognitive diversity reflects variation in developmental trajectories, not deviation from a singular norm. By shifting focus from correctional to contextual adaptation, research and practice can accommodate the full spectrum of human cognition. Continued interdisciplinary investigation will further clarify how early neural processes shape personality, learning and lived experiences across development.
Bibliography
Arnsten, A.F.T. (2009). The emerging neurobiology of attention deficit hyperactivity disorder: The key role of the prefrontal association cortex. The Journal of Pediatrics, 154(5), pp.I-S43.
Battle, D.E. (2013). Diagnostic and Statistical Manual of Mental Disorders (DSM). CoDAS, 25(2), pp.190–191.
Bourgeron, T. (2015). From the genetic architecture to synaptic plasticity in autism spectrum disorder. Nature Reviews Neuroscience, 16(9), pp.551–563.
Çetin, S., Uysal, S., Girgin, D., Alp, A., Kiliç, E. & Çiray, O. (2025). The Role of Neuroglia in Neurodevelopmental Disorders and Disruptive Behavior: A Broad Review of Current Literature. Neuroglia, 6(3), pp.34–34.
Courchesne, E., Campbell, K. & Solso, S. (2011). Brain growth across the life span in autism: Age-specific changes in anatomical pathology. Brain Research, 1380, pp.138–145.
Cundari, M., Vestberg, S., Gustafsson, P., Gorcenco, S. & Rasmussen, A. (2023). Neurocognitive and cerebellar function in ADHD, autism and spinocerebellar ataxia. Frontiers in Systems Neuroscience, 17, p.1168666.
De Silva, P.N. (2018). Do patterns of synaptic pruning underlie psychoses, autism and ADHD? BJPsych Advances, 24(3), pp.212–217.
Ecker, C., Bookheimer, S.Y. & Murphy, D.G.M. (2015). Neuroimaging in autism spectrum disorder: brain structure and function across the lifespan. The Lancet Neurology, 14(11), pp.1121–1134.
Eicher, J.D. & Gruen, J.R. (2014). Language Impairment and Dyslexia Genes Influence Language Skills in Children With Autism Spectrum Disorders. Autism Research, 8(2), pp.229–234.
Eltokhi, A., Janmaat, I.E., Genedi, M., Haarman, B.C.M. & Sommer, I.E.C. (2020). Dysregulation of synaptic pruning as a possible link between intestinal microbiota dysbiosis and neuropsychiatric disorders. Journal of Neuroscience Research, 98(7), pp.1335–1369.
Geschwind, D.H. & Levitt, P. (2007). Autism spectrum disorders: developmental disconnection syndromes. Current Opinion in Neurobiology, 17(1), pp.103–111.
Hutsler, J.J. & Zhang, H. (2010). Increased dendritic spine densities on cortical projection neurons in autism spectrum disorders. Brain Research, 1309, pp.83–94.
Huttenlocher, P.R. & Dabholkar, A.S. (1997). Regional differences in synaptogenesis in human cerebral cortex. The Journal of Comparative Neurology, 387(2), pp.167–178.
Jin, M., Xu, R., Wang, L., Mahabub Maraj Alam, Ma, Z., Zhu, S., Martini, A.C., Azadeh Jadali, Matteo Bernabucci, Xie, P., Kwan, K.Y., Pang, Z.P., Head, E., Liu, Y., Hart, R.P. & Jiang, P. (2022). Type-I-interferon signaling drives microglial dysfunction and senescence in human iPSC models of Down syndrome and Alzheimer’s disease. Cell stem cell, 29(7), pp.1135-1153.e8.
Johnson, M., Åsberg Johnels, J., Östlund, S., Cedergren, K., Omanovic, Z., Hjalmarsson, K., Jakobsson, K., Högstedt, J. & Billstedt, E. (2021). Long-term medication for ADHD and development of cognitive functions in children and adolescents. Journal of Psychiatric Research, 142(142), pp.204–209.
Khalid, U., Naeem, M., Stasolla, F., Syed, M., Abbas, M. & Coronato, A. (2024). Impact of AI-Powered Solutions in Rehabilitation Process: Recent Improvements and Future Trends. International Journal of General Medicine, 17, pp.943–969.
Levman, J. (2025). Factors Affecting Macro-Structural Development in the Cerebral Cortex: The Potential Role of Tissue Removal Through Pruning and Apoptosis. Biology, 14(12), p.1651.
Lord, C., Brugha, T.S., Charman, T., Cusack, J., Dumas, G., Frazier, T., Jones, E.J.H., Jones, R.M., Pickles, A., State, M.W., Taylor, J.L. & Veenstra-Vander Weele, J. (2020). Autism spectrum disorder. Nature Reviews Disease Primers, 6(1), pp.1–23.
Mannarelli, D., Pauletti, C., Missori, P., Trompetto, C., Cotellessa, F., Fattapposta, F. & Currà, A. (2023). Cerebellum’s Contribution to Attention, Executive Functions and Timing: Psychophysiological Evidence from Event-Related Potentials. Brain Sciences, 13(12), p.1683.
McLaughlin, K.A., Sheridan, M.A., Winter, W., Fox, N.A., Zeanah, C.H. & Nelson, C.A. (2014). Widespread Reductions in Cortical Thickness Following Severe Early-Life Deprivation: A Neurodevelopmental Pathway to Attention-Deficit/Hyperactivity Disorder. Biological Psychiatry, 76(8), pp.629–638.
Ntalindwa, T., Nduwingoma, M., Karangwa, E., Soron, T.R., Uworwabayeho, A. & Uwineza, A. (2021). Development of a Mobile App to Improve Numeracy Skills of Children With Autism Spectrum Disorder: Participatory Design and Usability Study. JMIR Pediatrics and Parenting, 4(3), p.e21471.
Stoodley, C.J. (2015). The Cerebellum and Neurodevelopmental Disorders. The Cerebellum, 15(1), pp.34–37.
Tang, G., Gudsnuk, K., Kuo, S.-H., Cotrina, Marisa L., Rosoklija, G., Sosunov, A., Sonders, Mark S., Kanter, E., Castagna, C., Yamamoto, A., Yue, Z., Arancio, O., Peterson, Bradley S., Champagne, F., Dwork, Andrew J., Goldman, J. & Sulzer, D. (2014). Loss of mTOR-Dependent Macroautophagy Causes Autistic-like Synaptic Pruning Deficits. Neuron, 83(5), pp.1131–1143.
Uddin, L.Q., Supekar, K. & Menon, V. (2013). Reconceptualizing functional brain connectivity in autism from a developmental perspective. Frontiers in Human Neuroscience, 7(458).
Ugarte, G., Piña, R., Contreras, D., Godoy, F., Rubio, D., Rozas, C., Zeise, M., Vidal, R., Escobar, J. & Morales, B. (2023). Attention Deficit-Hyperactivity Disorder (ADHD): From Abnormal Behavior to Impairment in Synaptic Plasticity. Biology, 12(9), p.1241.
Wang, L., Wang, B., Wu, C., Wang, J. & Sun, M. (2023). Autism Spectrum Disorder: Neurodevelopmental Risk Factors, Biological Mechanism, and Precision Therapy. International Journal of Molecular Sciences, 24(3), p.1819.
Zoghbi, H.Y. & Bear, M.F. (2012). Synaptic Dysfunction in Neurodevelopmental Disorders Associated with Autism and Intellectual Disabilities. Cold Spring Harbor Perspectives in Biology, 4(3), pp.a009886–a009886.